ABOUT MOCK RECALL
A mock recall in the pharmaceutical industry is a practice test to check how well a company can remove a medicine from the market if there is a problem with it. The company chooses a batch of medicine and acts as if it needs to recall it. They check where the batch was sent, how much was sold, and how quickly they can get it back. This helps the company find any problems in their system before a real recall happens. Mock recalls are important to make sure medicines are safe and that the company is ready in case of an emergency.
A Mock recall procedure shall be conducted once in a year & report for the same shall be maintained by QA.
Mock recall is limited only up to the level of collecting the product distribution information in Coordination with recall committee with a specified time frame.
Planner for Mock recall shall be prepared as per format as shown in format Mock Recall. Additional mock recall due to any reason shall be record in same.
For Mock Recall distribution detail shall be filled as per format Distribution Details.
Mock Product Recall Report shall be prepared as per format Mock Product Recall.
Information of the quantity of Batches laying the distribution chain shall be collected by QA/PPIC team for making effective product recall.
Rating of Evaluation Parameter:

Tracking Forward and Back Recall:
For traceability, parts and products are identified individually or by lots and information is accumulated in each process. Tracing forward means using accumulated information to track the movement of products and tracing back means tracking records (BMR/ BPR/ Material Movement in individual batch) backward in the timeline.
Tracing forward is an action to track a product by following the timeline. When a defect is detected in a particular part, batch or lot for instance, products containing the part can be identified to recall them precisely. Consequently, it is effective for the measures against recalls and defective products.
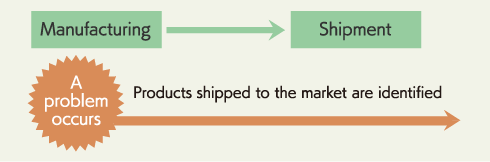
Tracking backward is an action to track records backward in the timeline. For example, when a problem occurs with shipped products, the relevant lot and process (Raw material, packing material detail) can be identified to investigate the cause promptly. Identification of a lot or a process allows quick actions to improve processes and quality, which leads to higher and more stable product quality.

For tracking backward investigation shall be carried out and during investigation manufacturing process, equipment, utility, man, material, method and mother nature shall be consider and based on investigation needed corrective and preventive action shall be taken. As per customer requirement investigation report shall be sent.
Recall to be done as per International guidelines and Global Pharmaceutical Regulatory Authorities.
Click the link for download a Word file copy of Recall Procedure:
https://pharmaguidehub.com/product/product-recall/
ANNEXURE FOR MOCK RECALL LETTER

ANNEXURE FOR DISTRIBUTION DETAIL


Click the link for download a Word file copy of Recall Procedure:
https://pharmaguidehub.com/product/product-recall/
ANNEXURE FOR MOCK PRODUCT RECALL REPORT




You Can Also Read:
STANDARD OPERATING PROCEDURE FOR INVESTIGATION IN PHARMACEUTICAL INDUSTRY
INVESTIGATION OF OUT OF SPECIFICATION (OOS)
STANDARD OPERATING PROCEDURE FOR THE HANDLING OF DEVIATIONS AND INCIDENTS





