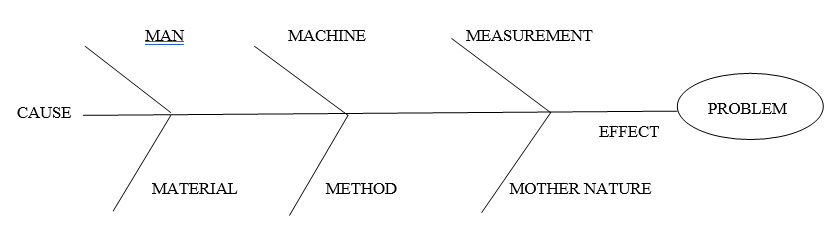
ABOUT FISHBONE DIAGRAM:
A fish bone diagram, also called an Ishikawa diagram, is a simple tool used in the pharmaceutical industry to find the causes of a problem. It looks like a fish skeleton, with the main problem written at the “head” and different causes shown along the “bones.” In pharma, it helps teams understand why issues like product defects, delays, or quality failures happen. Causes are often grouped into categories like methods, materials, machines, and people. By using the fish bone diagram, teams can work together to find the root cause of problems and fix them. It is a very helpful tool.
Fish Bone Diagram (Cause and Effect Diagram)
Man: This category takes in to consideration the human element that may affect the problem. It considers the people that touch the process.
If the person s involved in activity is not trained.
If new person involved in activity.
If person is competent for the activity.
Relevant experience of the concern personnel.
Material: This category takes into the consideration the substances out of which the product can be made. It considers all material that touches the process.
If material is not appropriate.
Any change in vendor
Vendor is not qualified
Change in specification / grade or material
Machine: This category takes in to the consideration the equipment used that may affect the problem. It considers the equipment that touches the product, process and system.
If periodic Preventive maintenance of machine not done.
If any breakdown has occurred during process.
If machine Qualification/ Requalification done.
If instrument /Equipment showing actual reading.
If machine setting done between the activity.
Method: This category takes in to the consideration the method/ process that may affect the problem
If any change in machine process and product parameter.
If batch processed as per written instructions mentioned in Batch Manufacturing record.
Measurement: This category takes in to the consideration measurement that may affect the problem. Measurement considers the measurements used for the product, process system.
Calibration of equipment/Instruments.
Mother Nature / Environment: This category takes in to the consideration the Environmental factors that may affect the problem. It considers the environmental factor that touches the process.
Investigate with sufficient depth so as to identify the root cause / probable root cause. The examples of possible causes are given below:
- Personnel not adequately trained
- Procedures inadequate
- Personnel trained but did not follow standard operating procedure (trained but forgot, working too fast, took shortcuts and used wrong procedures)
- Equipment used not qualified
- Area cleaning / equipments cleaning not adequate
- Error in environmental monitoring
- Error in Preventive Maintenance
- Error in calibration
Investigation shall be done by review of concerned documents i.e. BMR, BPR, logbooks, relevant SOP’s, analytical data, raw data, stability data, preventive and breakdown maintenance data, calibration records, validation reports and any other relevant documents shall be reviewed to collect maximum possible and relevant data.
If required necessary interrogation shall be done up to doer/performer level to find out the route cause.
After going through all relevant documents and data collected through the discussions with relevant personnel, Investigation team will suggest the next step such as resampling, retesting, extra testing and any other measure required (wherever applicable) to complete the investigation.
However, any Resampling, retesting or extra testing required shall be approved by Head QA.
Based upon investigation outcome, team shall identify the root cause of the non conformance.
After identification of root cause of the problem Impact/Risk assessment shall be performed as per SOP, wherever applicable.
Designated person from QA shall confirm the authenticity and completeness of the data and analyze the data to identify potential product and quality problems for which corrective action and preventive actions are required.
CAPA shall be decided based on scientific and logical justifications to prevent future reoccurrence of the similar problem.
You Can Also Read:
STANDARD OPERATING PROCEDURE FOR INVESTIGATION IN PHARMACEUTICAL INDUSTRY
INVESTIGATION OF OUT OF SPECIFICATION (OOS)
STANDARD OPERATING PROCEDURE FOR THE HANDLING OF DEVIATIONS AND INCIDENTS