Validation Master Plan (VMP) describes the company’s intentions regarding the validation of equipments, instruments, systems, utilities, facilities, materials, analytical methods and processes.
Validation Master Plan, being a dynamic document, presents a current overview of the entire validation activities, its organizational structure and planning for the current year.
Validation Master Plan ensures that:
VMP adds value to our quality system and provides assurance on the product quality, by ensuring that our facility, equipment and processes remain in a state of validation.
A suitable validation program is designed, in order to demonstrate that the facility is capable of meeting process / product parameters in a repeatable and controllable manner.
The facility is designed to provide the desired degree of environment controls as well as manufacturing controls, for all the production steps and throughout the manufacturing process of the Pharmaceutical Products.
The critical programs for utilities qualification, process validation and equipment qualification have been established, in accordance with the product requirements (based on the product information and the cGMP guidelines and other regulations).

Key Parameter of Below Page
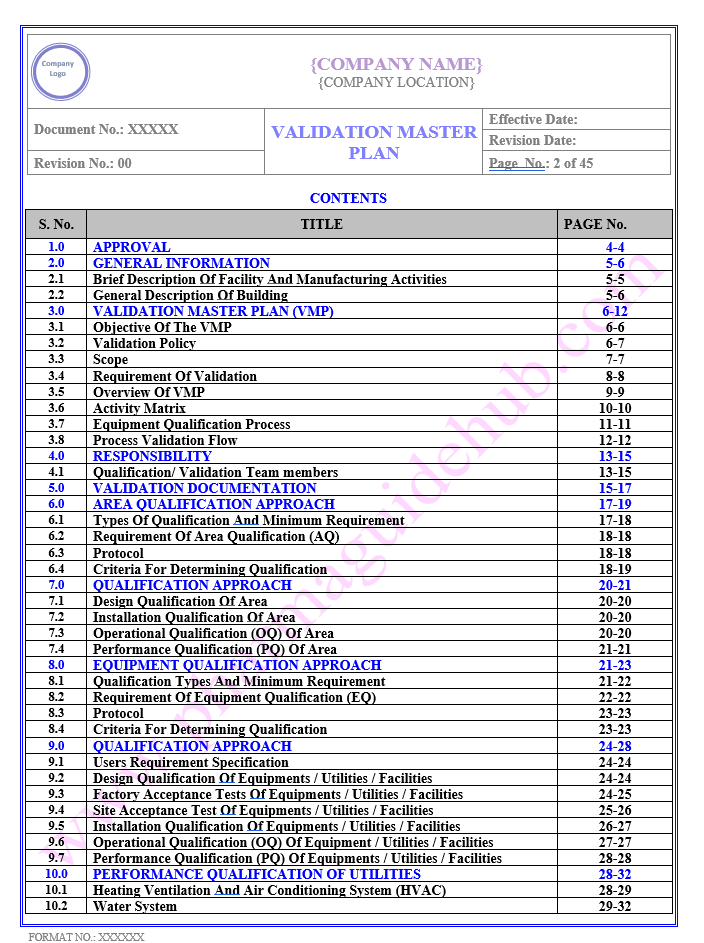
TABLE OF CONTENTS
Find below pages for complete Manual
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/validation-master-plan/
Key Parameter of Below Page
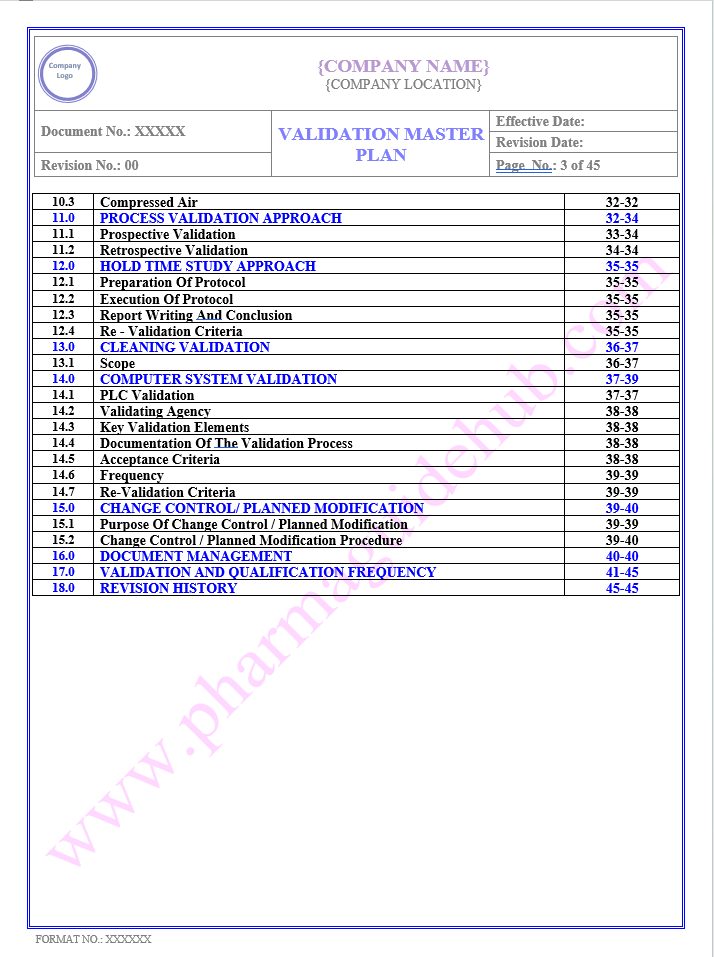
TABLE OF CONTENTS
Find below pages for complete Manual
Key Parameter of Below Page
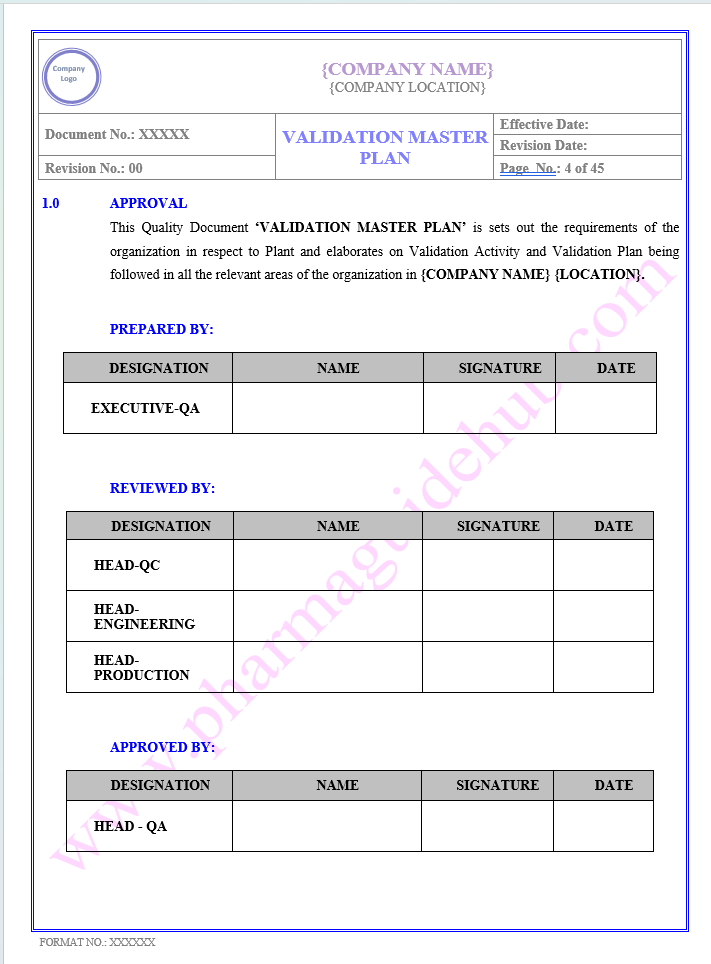
APPROVAL
Find below pages for complete Manual
Key Parameter of Below Page
GENERAL INFORMATION
Find below pages for complete Manual

Key Parameter of Below Page
TABLE OF CONTENTS
VALIDATION MASTER PLAN
Find below pages for complete Manual

Key Parameter of Below Page
VALIDATION MASTER PLAN
Find below pages for complete Manual
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/validation-master-plan/

Key Parameter of Below Page
VALIDATION MASTER PLAN
Find below pages for complete Manual

Key Parameter of Below Page
VALIDATION MASTER PLAN
Find below pages for complete Manual
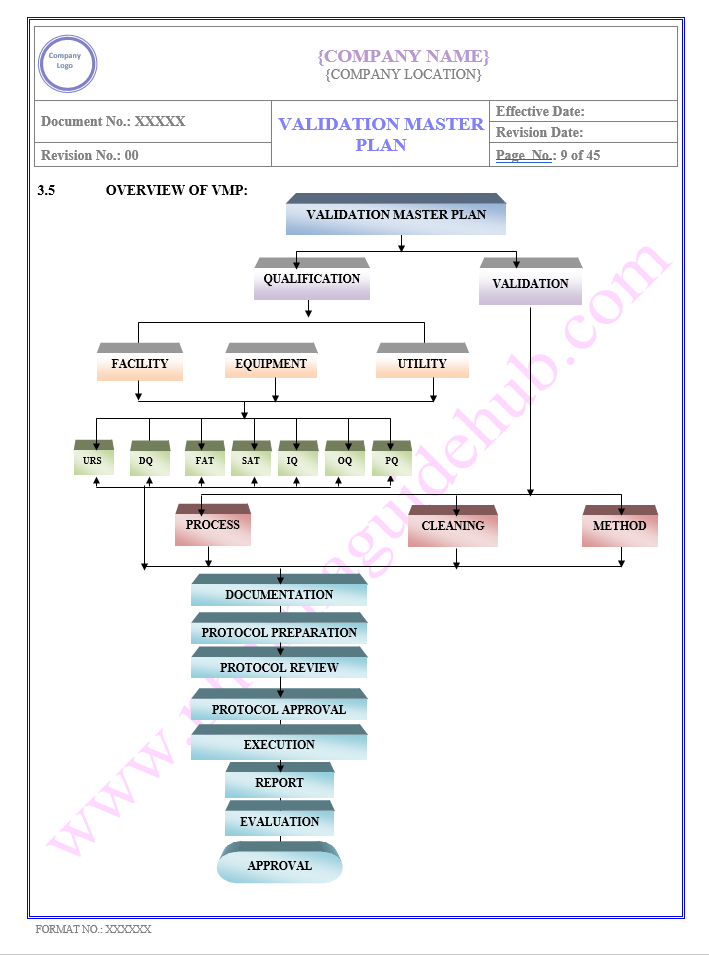
Key Parameter of Below Page
VALIDATION MASTER PLAN
Find below pages for complete Manual
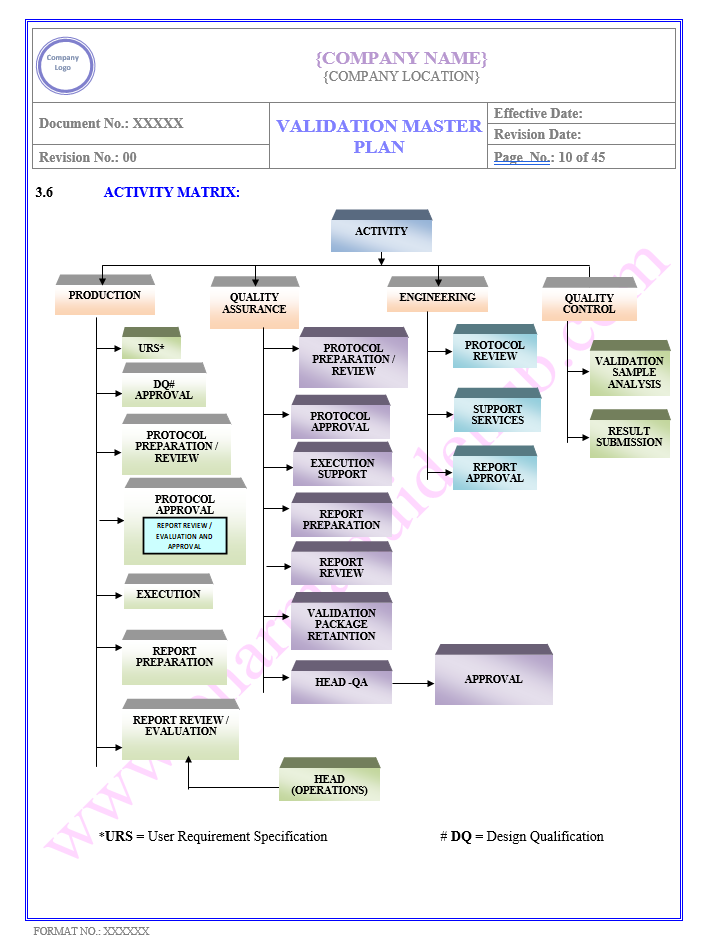
Key Parameter of Below Page
VALIDATION MASTER PLAN
Find below pages for complete Manual
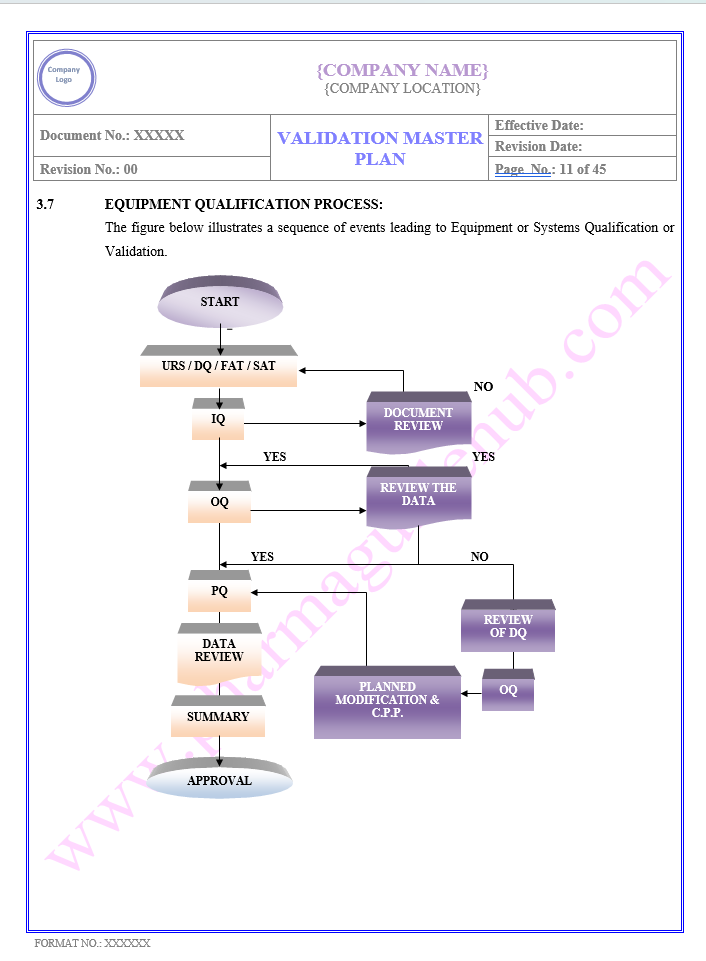
Key Parameter of Below Page
VALIDATION MASTER PLAN
Find below pages for complete Manual
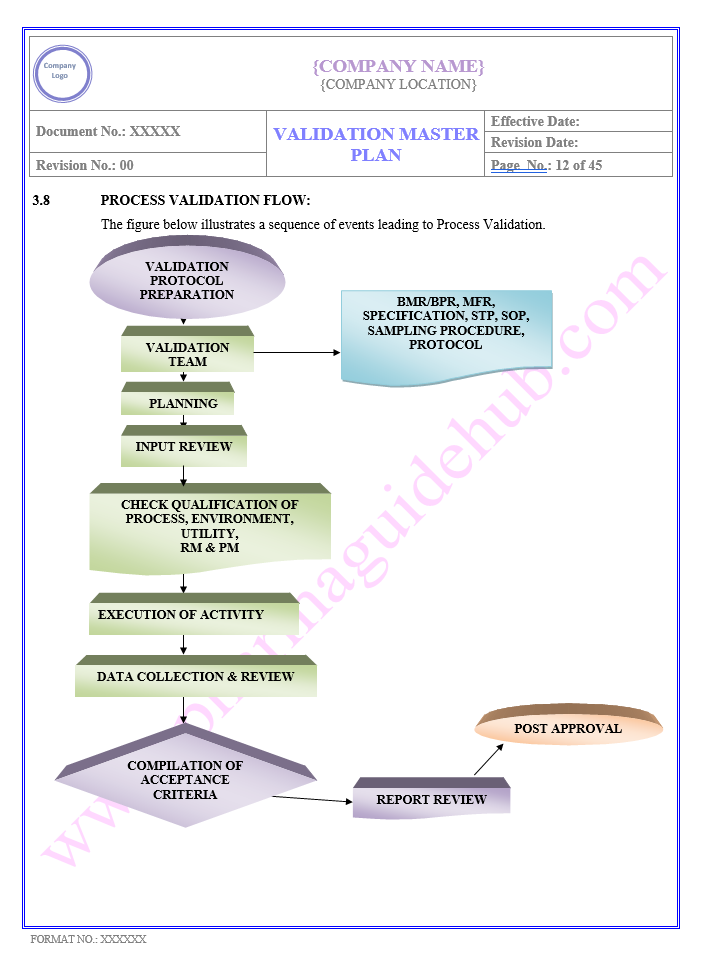
Key Parameter of Below Page
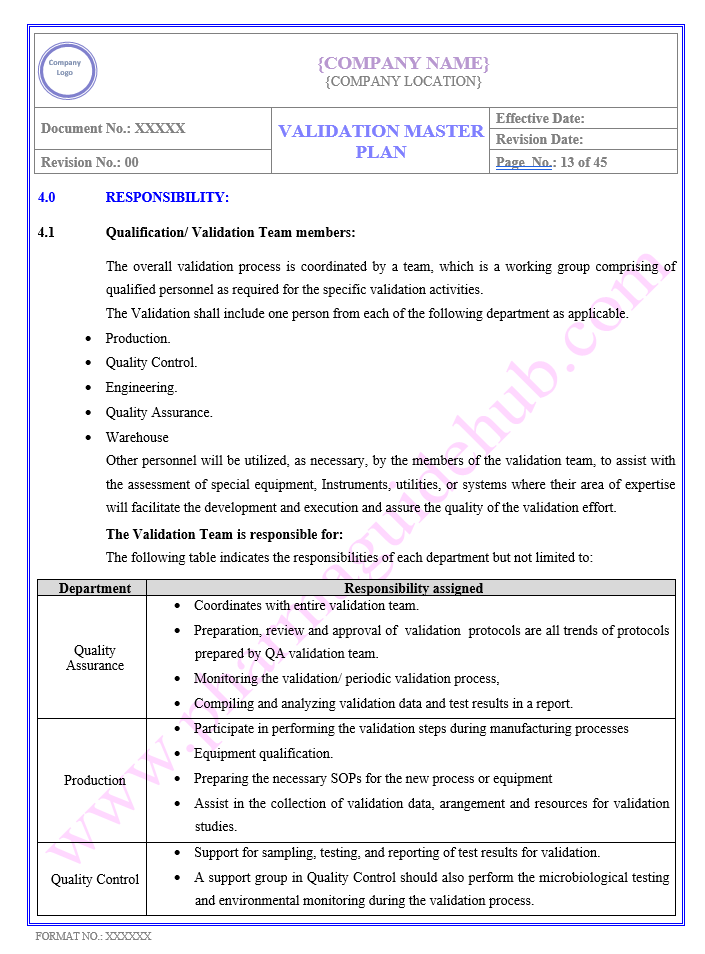
RESPONSIBILITY
Find below pages for complete Manual
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/validation-master-plan/
Key Parameter of Below Page
RESPONSIBILITY
Find below pages for complete Manual

Key Parameter of Below Page
RESPONSIBILITY
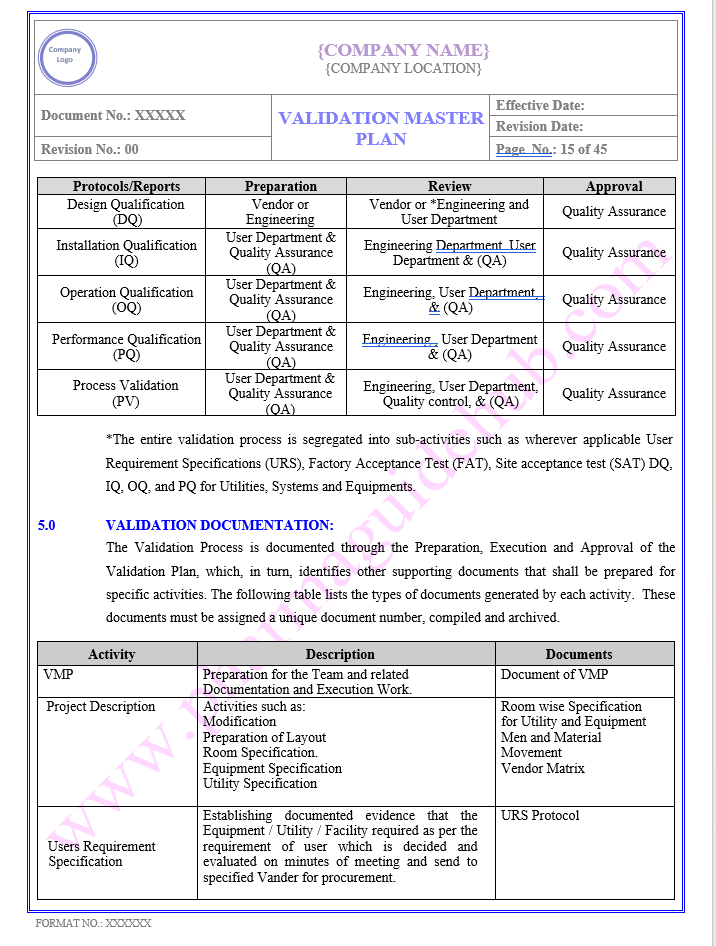
VALIDATION DOCUMENTS
Find below pages for complete Manual
Key Parameter of Below Page
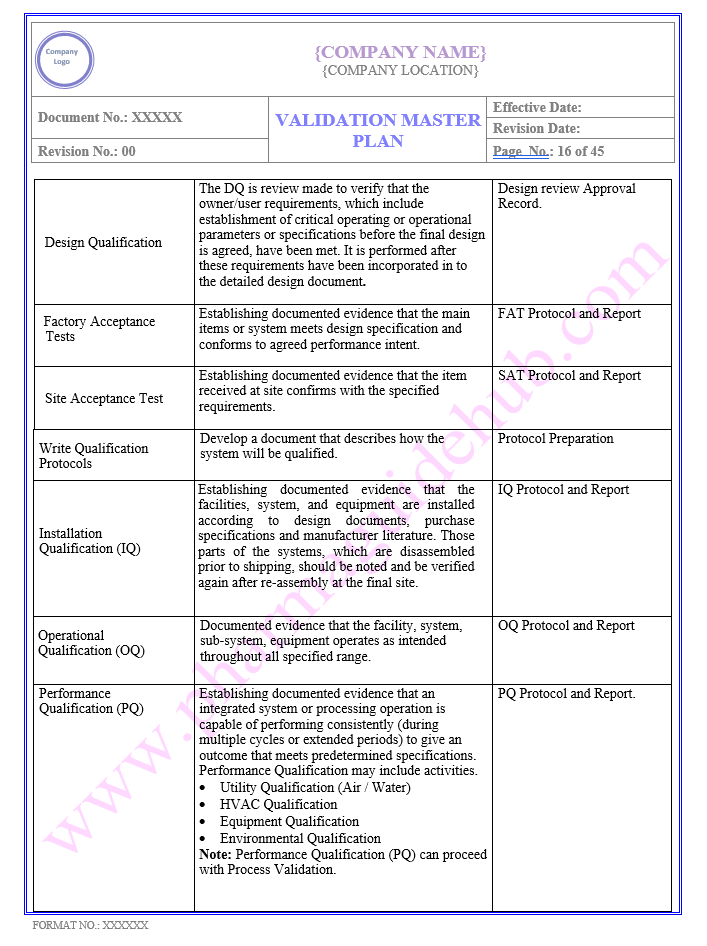
VALIDATION DOCUMENTS
Find below pages for complete Manual
Key Parameter of Below Page
VALIDATION DOCUMENTS
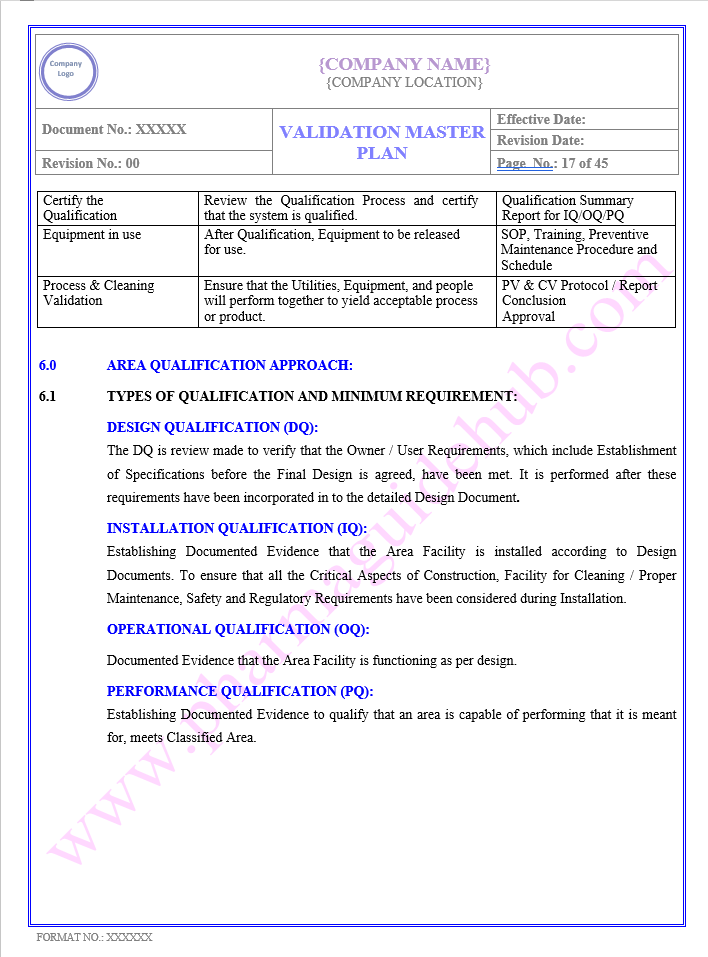
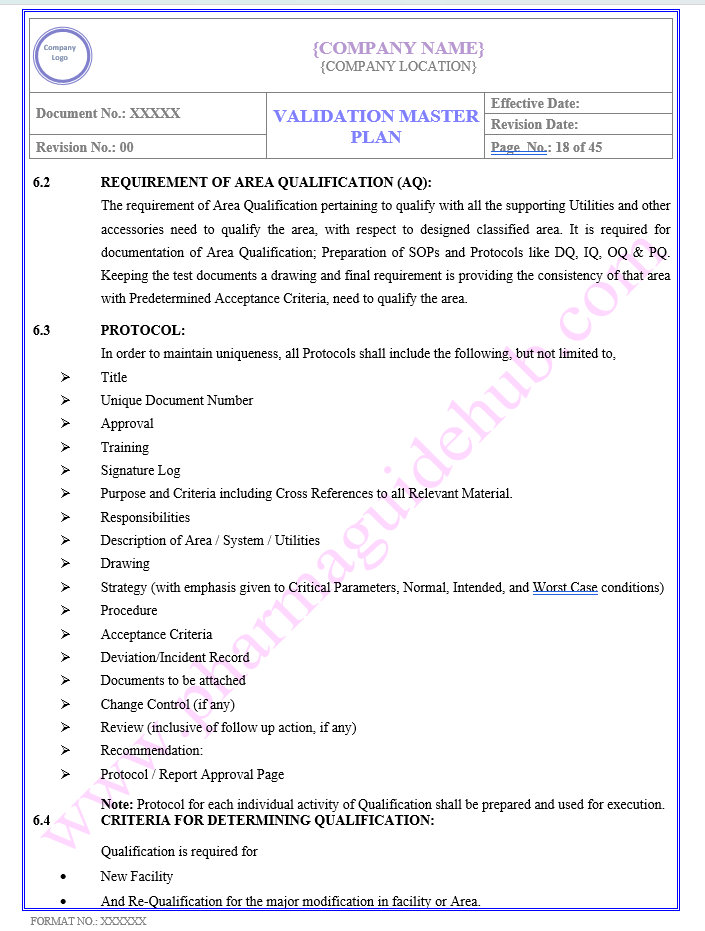
AREA QUALIFICATION APPROACH
Find below pages for complete Manual
Key Parameter of Below Page
AREA QUALIFICATION APPROACH
Find below pages for complete Manual
Key Parameter of Below Page
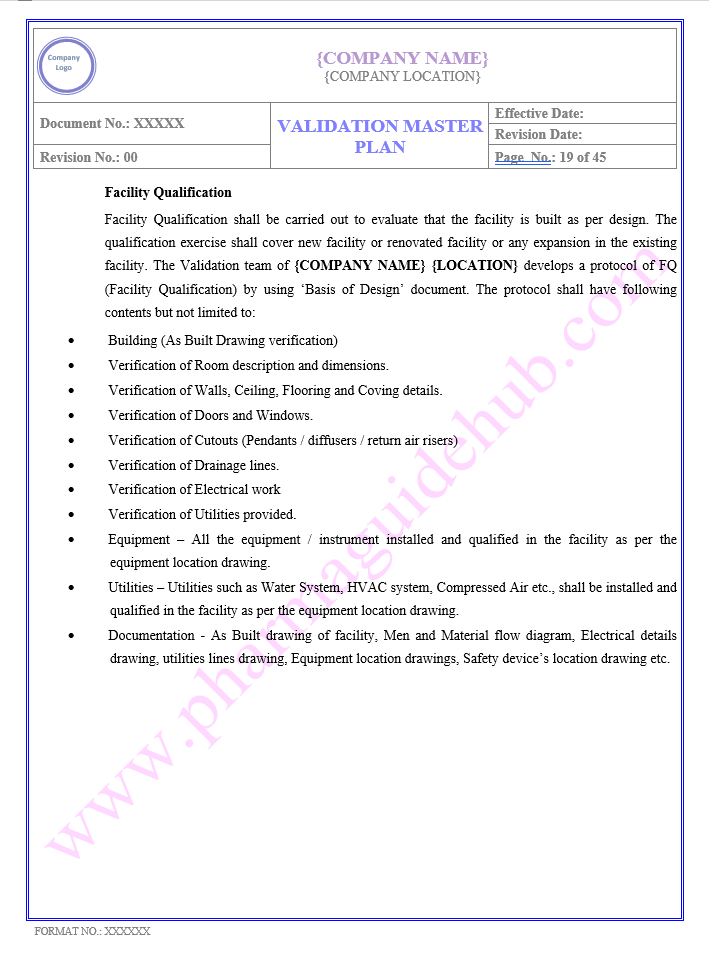
AREA QUALIFICATION APPROACH
Find below pages for complete Manual
Key Parameter of Below Page
QUALIFICATION APPROACH
Find below pages for complete Manual
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/validation-master-plan/

Key Parameter of Below Page
QUALIFICATION APPROACH
EQUIPMENT QUALIFICATION APPROACH
Find below pages for complete Manual

Key Parameter of Below Page
EQUIPMENT QUALIFICATION APPROACH
Find below pages for complete Manual

Key Parameter of Below Page
EQUIPMENT QUALIFICATION APPROACH
Find below pages for complete Manual

Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/validation-master-plan/