During the WHO GMP inspection of a pharmaceutical plant, Name AABBCC, an observation was made that the QUALITY MANUAL WAS NOT UPDATED AS PER CURRENT REGULATORY REQUIREMENTS. This has been received.
The compliance report along with supporting documents is to be submitted within 30 days.
Context of Observation:
During the WHO-GMP inspection, it was observed that although a Quality Manual is available at the site, it has not been updated to reflect current regulatory requirements, including recent WHO-GMP guidelines and internal process updates.
The inspection team noted that:
- The Quality Manual version in use is outdated.
- Recent changes in quality systems (e.g., CAPA, data integrity, risk management) are not reflected.
- Lack of alignment between actual practices and documented Quality Manual.
This indicates a gap in document control and regulatory compliance, potentially impacting the effectiveness of the Quality Management System (QMS).
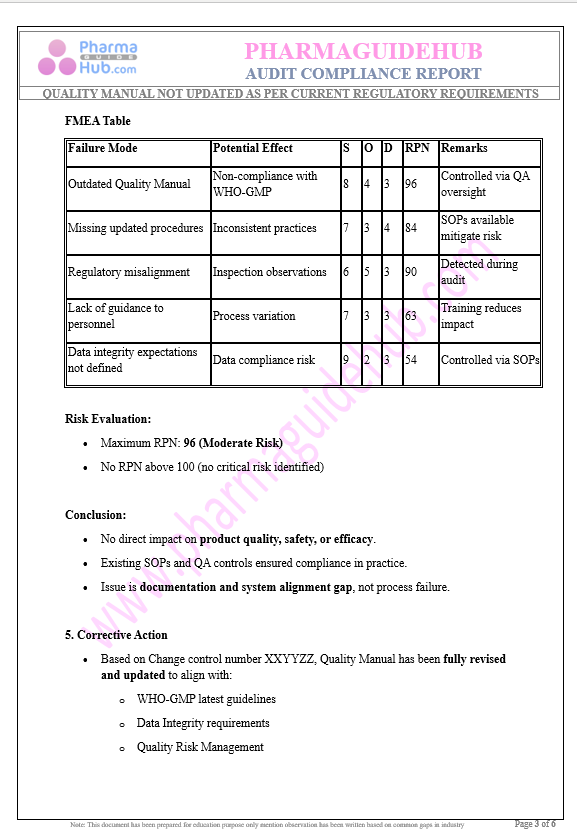
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-quality-manual-not-updated-as-per-current-regulatory-requirements/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-quality-manual-not-updated-as-per-current-regulatory-requirements/

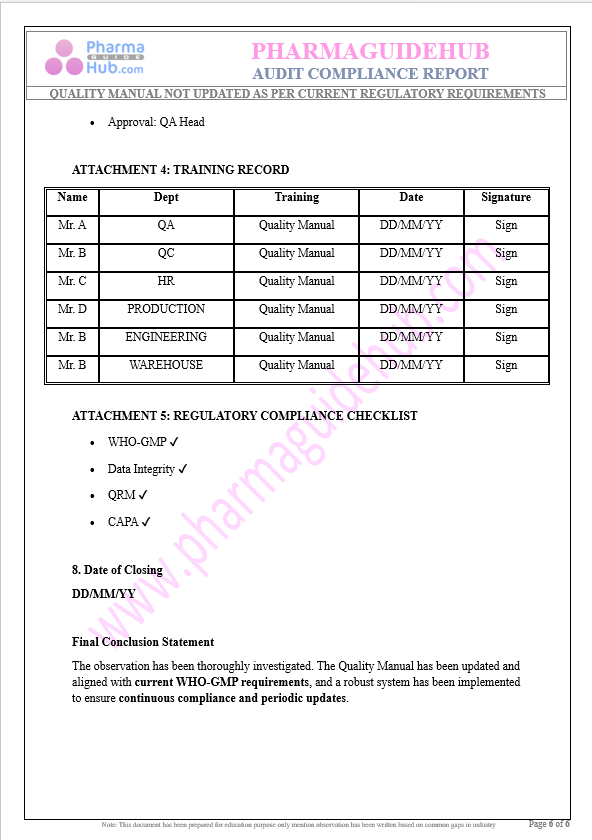
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-quality-manual-not-updated-as-per-current-regulatory-requirements/





