A training manual in the pharmaceutical industry serves as a comprehensive guide for employees, outlining procedures, regulations, and best practices crucial for their roles. It ensures consistency and compliance across operations, covering topics like Good Manufacturing Practices (GMP), Standard Operating Procedures (SOPs), equipment handling, and safety protocols. These manuals are vital for onboarding new staff, providing ongoing training, and facilitating knowledge retention. They are meticulously developed and regularly updated to reflect the latest regulatory requirements and industry standards, ultimately contributing to product quality, patient safety, and operational efficiency within the pharmaceutical sector.
Part 2 of 2
Click the link for part 1 of 2:
https://pharmaguidehub.com/training-manual/

Key Parameter of Below Page
BASICS OF CGMP AND INTRODUCTION TO SCHEDULE-M
Find below pages for complete protocol:

Key Parameter of Below Page
BASICS OF CGMP AND INTRODUCTION TO SCHEDULE-M
Find below pages for complete protocol:

Key Parameter of Below Page
BASICS OF CGMP AND INTRODUCTION TO SCHEDULE-M
Find below pages for complete protocol:
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/training-manual/

Key Parameter of Below Page
BASICS OF CGMP AND INTRODUCTION TO SCHEDULE-M
Find below pages for complete protocol:

Key Parameter of Below Page
VALIDATION
Find below pages for complete protocol:

Key Parameter of Below Page
VALIDATION
Find below pages for complete protocol:

Key Parameter of Below Page
VALIDATION
Find below pages for complete protocol:

Key Parameter of Below Page
VALIDATION
Find below pages for complete protocol:

Key Parameter of Below Page
VALIDATION
Find below pages for complete protocol:

Key Parameter of Below Page
PACKING AND LABELLING
Find below pages for complete protocol:
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/training-manual/

Key Parameter of Below Page
PACKING AND LABELLING
Find below pages for complete protocol:

Key Parameter of Below Page
PACKING AND LABELLING
Find below pages for complete protocol:

Key Parameter of Below Page
PACKING AND LABELLING
Find below pages for complete protocol:

Key Parameter of Below Page
PACKING AND LABELLING
Find below pages for complete protocol:

Key Parameter of Below Page
BASIC OF GLP AND ROLE OF QUALITY CONTROL DEPARTMENT
Find below pages for complete protocol:

Key Parameter of Below Page
BASIC OF GLP AND ROLE OF QUALITY CONTROL DEPARTMENT
Find below pages for complete protocol:

Key Parameter of Below Page
BASIC OF GLP AND ROLE OF QUALITY CONTROL DEPARTMENT
Find below pages for complete protocol:

Key Parameter of Below Page
BASIC OF GLP AND ROLE OF QUALITY CONTROL DEPARTMENT
Find below pages for complete protocol:

Key Parameter of Below Page
SELF INSPECTION
Find below pages for complete protocol:

Key Parameter of Below Page
COMPLAINTS, RECALLS AND WITHDRAWALS
Find below pages for complete protocol:
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/training-manual/

Key Parameter of Below Page
COMPLAINTS, RECALLS AND WITHDRAWALS
Find below pages for complete protocol:
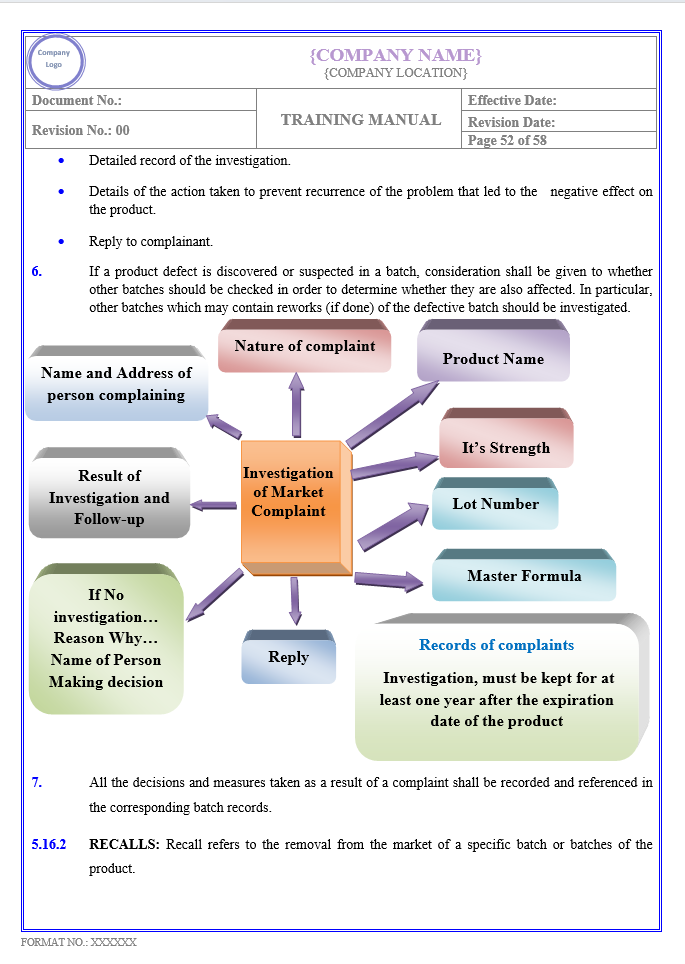
Key Parameter of Below Page
COMPLAINTS, RECALLS AND WITHDRAWALS
GLOSSARY
Find below pages for complete protocol:

Key Parameter of Below Page
GLOSSARY
Find below pages for complete protocol:

Key Parameter of Below Page
GLOSSARY
Find below pages for complete protocol:

Key Parameter of Below Page
GLOSSARY
Find below pages for complete protocol:
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/training-manual/

Key Parameter of Below Page
GLOSSARY
Find below pages for complete protocol:

Key Parameter of Below Page
REVISION HISTORY
Find below pages for complete protocol:
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/training-manual/




