OBJECTIVE:
To lay down a Procedure for writing the Standard Operating procedure for stack height monitoring of finished goods shippers.
SCOPE:
This SOP is applicable for the transportation study of first three batches of all modes of route of transportation for specified container to know the stack height of finished goods shipper at FG store and during transportation at {Company Name} {Location}.
RESPONSIBILITY:
Concerned department (Operating Executive/ Designee) shall be responsible for preparation of SOPs.
Operating Manager / Designee shall be responsible for review of SOPs.
QA Manager / Designee shall be responsible for Approval of SOPs.
ACCOUNTABILITY:
QA Manager Shall is accountable for implementation of SOPs.
ABOUT STACK HEIGHT
Stack height monitoring of finished goods shippers in the pharmaceutical industry is crucial to ensure product integrity during storage and transportation. Excessive stack height can cause compression damage, compromising packaging and potentially affecting product quality. Monitoring systems help maintain compliance with Good Distribution Practices (GDP) by ensuring that stacking does not exceed manufacturer-recommended limits. Accurate stack height control also improves warehouse safety and space optimization. Technologies like load sensors, automated palletizers, and camera-based systems are often used for real-time monitoring. Overall, maintaining proper stack height safeguards pharmaceutical products, reduces waste, and ensures patient safety by preserving drug efficacy and packaging integrity.
PROCEDURE:
DROP TEST PROCEDURE FOR STACK HIGHT MONITORING:
The purpose of the test is to evaluate the stack height during handling of the shippers containing the drug products in such a manner as to prevent damage to the product during packing, storage, transit and handling. These containers should be subjected to a drop test to prove the packaging is adequate.
NOTE: Any flat surface, such as a table top, may be used if set at the prescribed height, dropping package onto a hard, flat floor
Identify the surface as follows:
1. Top
2. Small side
3. Base
4. near side
5. Largest Side
6. Smallest side
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/stack-height-monitoring-of-finished-goods-shippers/
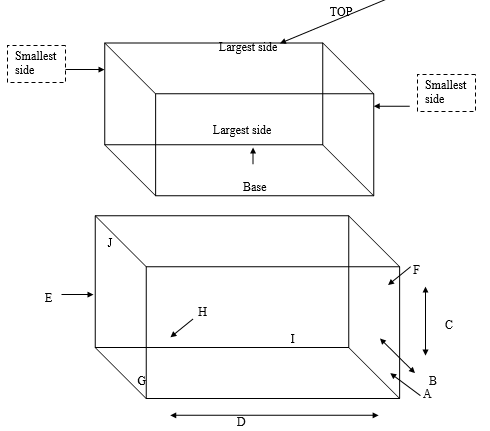
Drop package from pre-determined height in the following order (do not throw).
A. Corner
B. Shortest edge radiating horizontally from corner A
C. Next shortest edge radiating from same corner
D. Longest edge radiating vertically from corner A H
E. Flat on one of smallest sides
F. Flat on opposite smallest side
G. Flat on one base
H. Flat on top
I. Flat on one largest side
J. Flat on opposite largest side
Initial set the height for 6 filled shipper layers for 7 ply shipper and 5 filled shipper layers for 5 ply shipper and record the observation in Annexure-I. Based on shipper size, the height of filled shipper layer shall vary. If the shipper size is small, then the number of layers can be increased and if shipper size is large then number of layers can be decrease.
Before start the stack height study ensure the placed maximum number of filled shippers in column wise shall remain in standing position without any support. The shipper column shall not swing if it’s found single column of filled shipper is swinging then number of layers can be decrease and record the observation in Annexure-I.
Kept the shipper for 30 (+10) days. After completion of 15 days filled shipper placed at bottom shall be inspected and any damage of content shall be recorded in the protocol cum report of drop test in Annexure-I. If any damage is observed within 15 days, then test shall be performed again to reduce the 1or more layer.
After completion of 30 (+10) days a drop test shall be performed and after completion of the 10 drops of top shipper, open package and inspect the shipper and content for damage and record in Annexure-I. If observed percentage of damage is more than acceptance limit, then test shall be performed again to reduce the 1or more layer.
For acceptance criteria of damage shall be followed as per mentioned table.
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/stack-height-monitoring-of-finished-goods-shippers/
| Sr. No. | Defected content | Category of defect | Acceptance Criteria | |
| 1. | Bottle/Carton/Monocarton/Cylindrical Box/Zip Pouch | Critical | 0% | |
| 2. | Box | Major | 3% | |
| 3. | Label | Critical | 0% | |
| 4. | Shrink | Minor | 5% | |
| 5. | Product | Critical | 0% | |
| 6. | Shipper | Minor Crack | Minor | 5% |
| Torn of minor edges | Major | 3% | ||
| Torn of surface | Critical | 0% | ||
| 7. | Seal damage | Major | 3% | |
The drop test should be performed once for all sides as per step no. 5.1.1 and shall be recorded in Annexure -I.
After completion of the drop test measure the height and record in protocol cum report of drop test in Annexure-I Based on the drop test report number of filled shipper shall be kept according to height as per summarized stack height.
Products which have same weight of content, same packing material and same packing style then based on equivalency study shall be contain same shipper stack height. In this case a separate stack height monitoring study is not required and same shall be evaluated through Annexure-I
PROTOCOL CUM REPORT CONTENTS FOR STACK HEIGHT MONITORING:
Protocol cum report pre approval
Introduction
Objective
Scope
Responsibility
Reason for filled shipper stack height monitoring.
Training record of concerned personnels
Procedure for stack height monitoring
Product detail
Observation after staging the filled shipper.
Equivalence study for filled shipper stack height monitoring.
References
Non conformance
Deviation
Change control if any
Review (inclusive of follow up action)
Conclusion
Recommendation
Abbreviation
Protocol cum report post approval
Revision history
REFERANCE:
In House
ANNEXURES:
| ANNEXURE No. | TITLE OF ANNEXURE |
| Annexure-I | Protocol cum report for filled shipper stack height monitoring |
ENCLOSURES: SOP Training Record.
DISTRIBUTION:
| Controlled Copy No. 01Controlled Copy No. 02 | : : | Manager Quality Assurance Warehouse Department |
| Master Copy | : | Quality Assurance Department |
ABBREVIATIONS:
| SOP | : | Standard Operating Procedure |
| No. | : | Number |
| QA | : | Quality Assurance |
| % | : | Percentage |
REVISION HISTORY:
CHANGE HISTORY LOG
| Revision No. | Details of Changes | Reason for Change | Effective Date |
| 00 | New SOP | Not Applicable | To Be Written Manual |
ANNEXURE-I

Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/stack-height-monitoring-of-finished-goods-shippers/





