Part 1 of 3
Quality Risk Management (QRM) in the pharmaceutical industry is a systematic process of identifying, assessing, controlling, and reviewing risks to the quality of drug products across their lifecycle. It’s a crucial element of Good Manufacturing Practices (GMP), ensuring patient safety and product efficacy. QRM employs tools like FMEA and HACCP to proactively identify potential hazards in manufacturing, supply chain, and distribution. By evaluating the probability and severity of these risks, companies can implement mitigation strategies, including CAPA and revised SOPs. Effective QRM fosters data-driven decision-making, enhances communication among stakeholders, and ensures continuous improvement through regular risk reviews, ultimately safeguarding product quality and regulatory compliance.

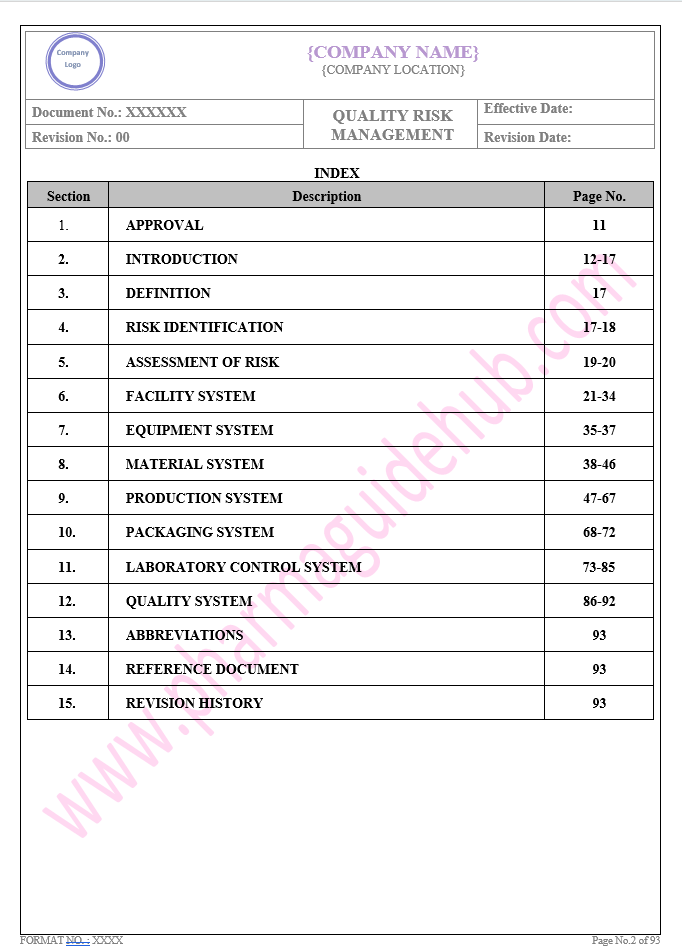
Key Parameter of Below Page:
Table of contents
Find the below page for content
Key Parameter of Below Page:
General
Find the below page for content

Key Parameter of Below Page:
General
Find the below page for content
Click the link to download complete word file copy of this document:
https://pharmaguidehub.com/product/quality-risk-management/

Key Parameter of Below Page:
General
Find the below page for content
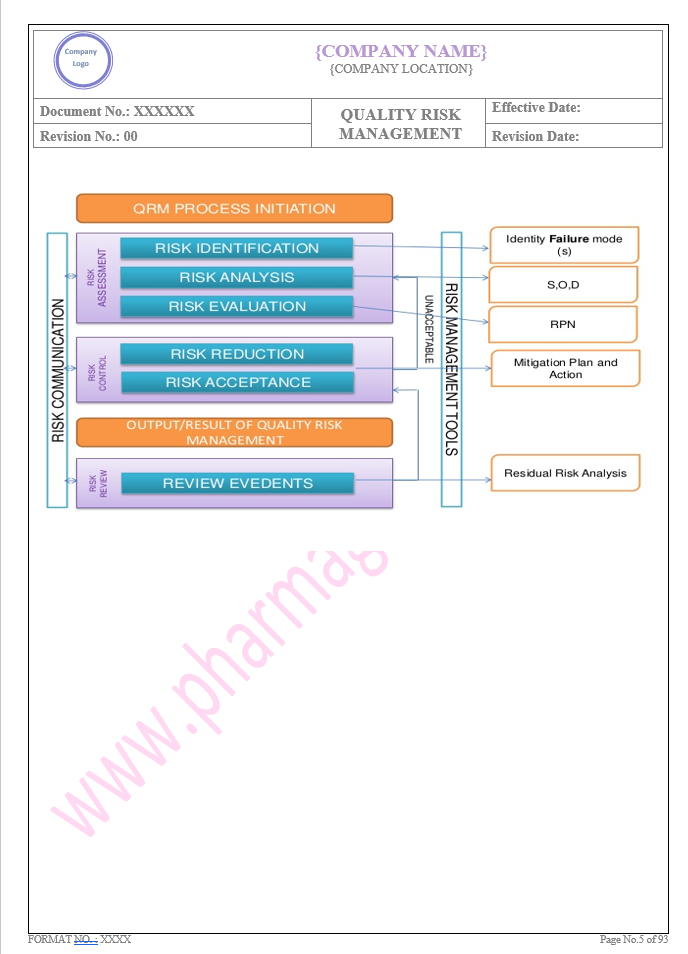
Key Parameter of Below Page:
General
Find the below page for content

Key Parameter of Below Page:
General
Find the below page for content

Key Parameter of Below Page:
General
Find the below page for content
Click the link to download complete word file copy of this document:
https://pharmaguidehub.com/product/quality-risk-management/

Key Parameter of Below Page:
General
Find the below page for content

Key Parameter of Below Page:
General
Find the below page for content

Key Parameter of Below Page:
Approval
Find the below page for content
Key Parameter of Below Page:
Introduction
Find the below page for content

Key Parameter of Below Page:
Introduction
Find the below page for content
Click the link to download complete word file copy of this document:
https://pharmaguidehub.com/product/quality-risk-management/
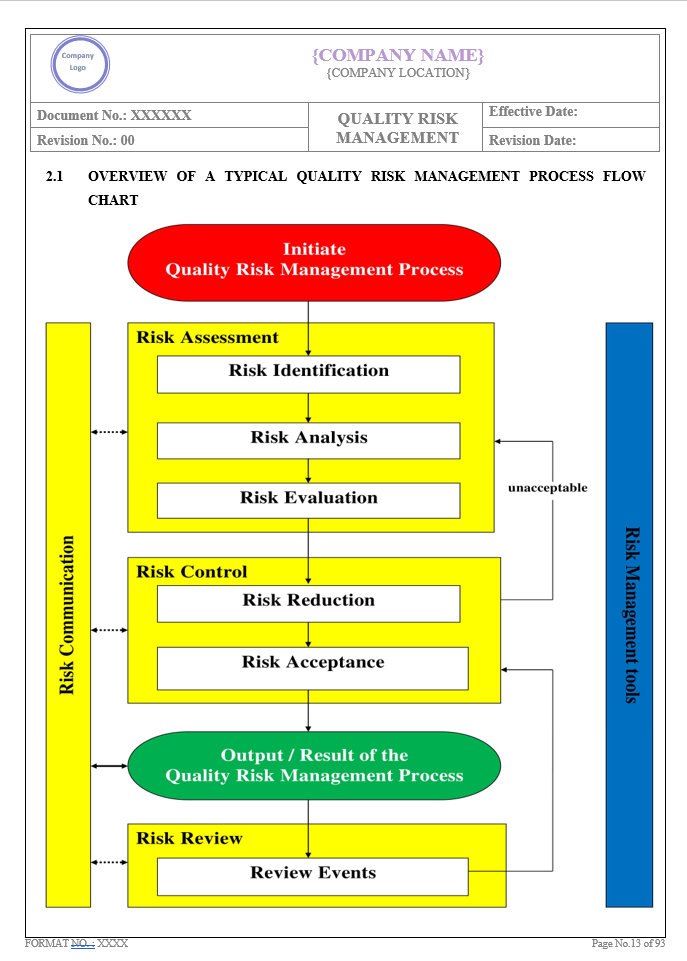
Key Parameter of Below Page:
Introduction
Find the below page for content

Key Parameter of Below Page:
Introduction
Find the below page for content
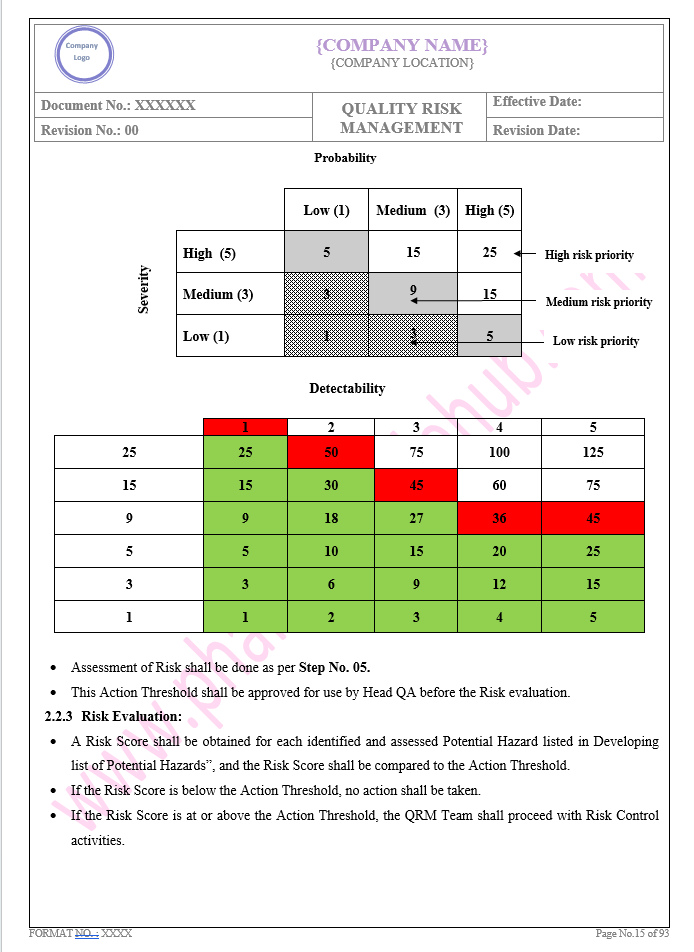
Key Parameter of Below Page:
Introduction
Find the below page for content

Key Parameter of Below Page:
Introduction
Definition
Find the below page for content
Click the link to download complete word file copy of this document:
https://pharmaguidehub.com/product/quality-risk-management/

Key Parameter of Below Page:
Definition
Risk Identification
Find the below page for content

Key Parameter of Below Page:
Assessment of Risk
Find the below page for content
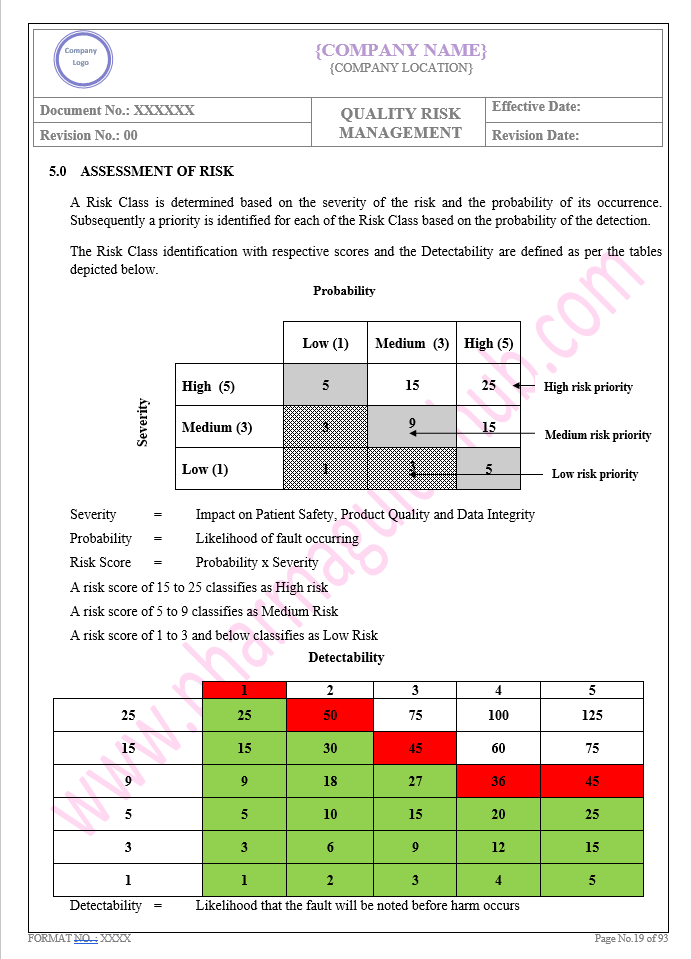
Key Parameter of Below Page:
Assessment of Risk
Find the below page for content
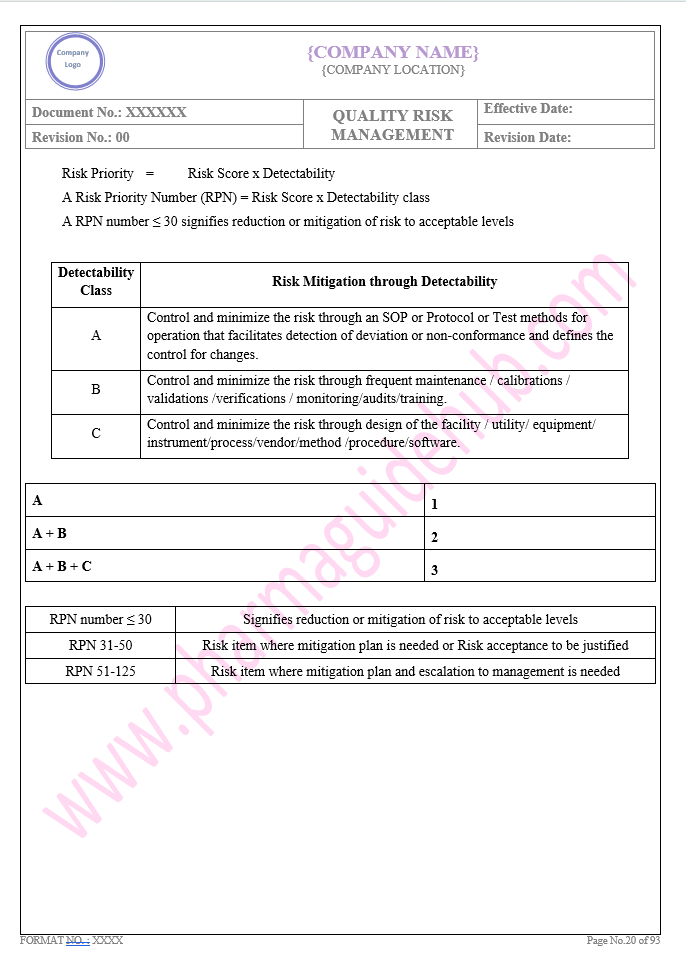
Key Parameter of Below Page:
Facility system
Find the below page for content
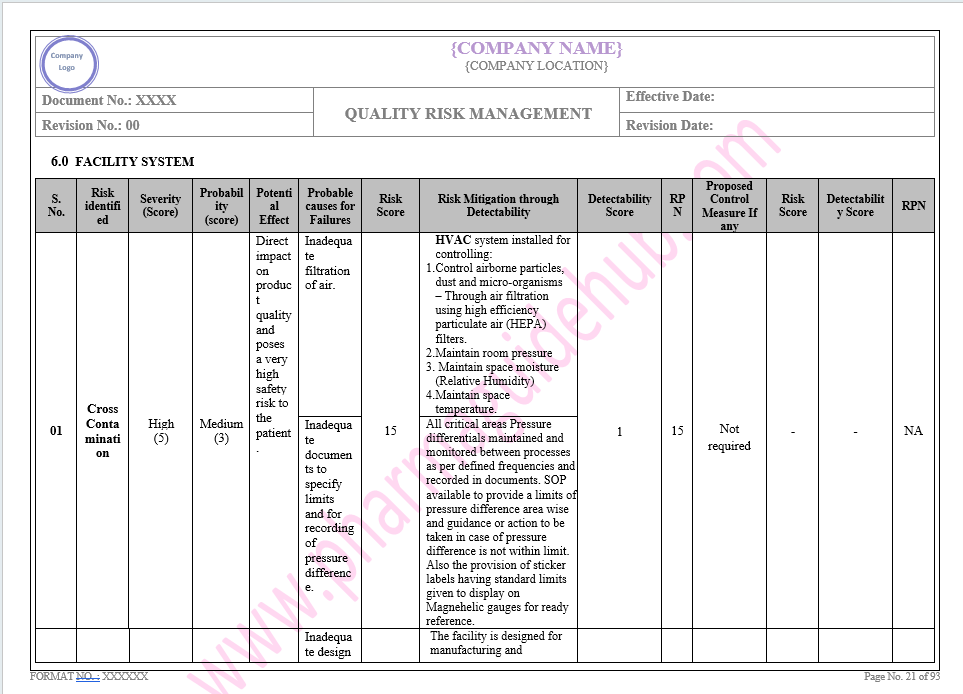
Key Parameter of Below Page:
Facility system
Find the below page for content
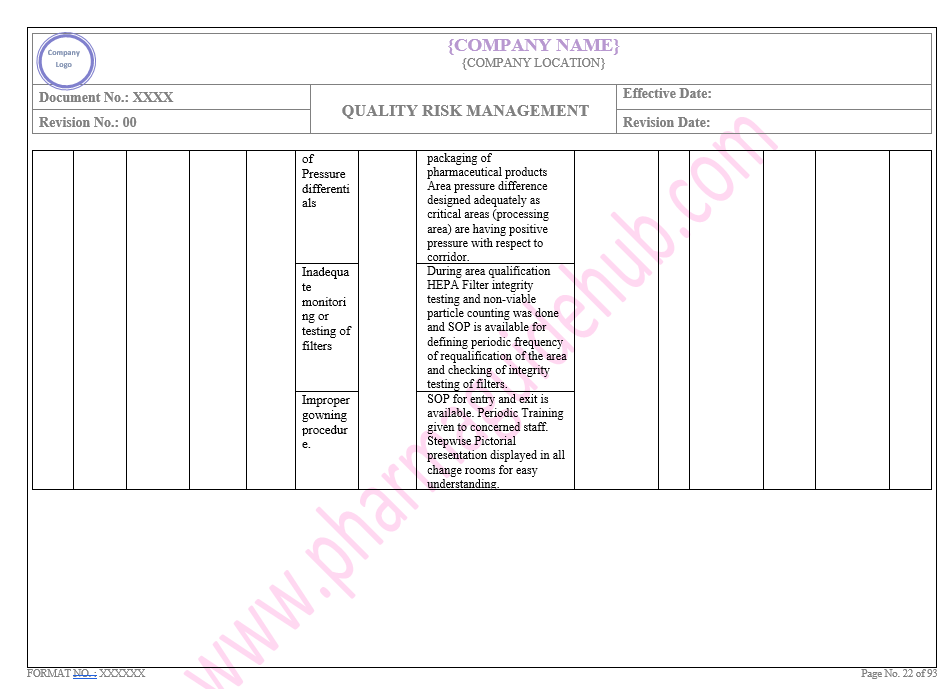
Key Parameter of Below Page:
Facility system
Find the below page for content
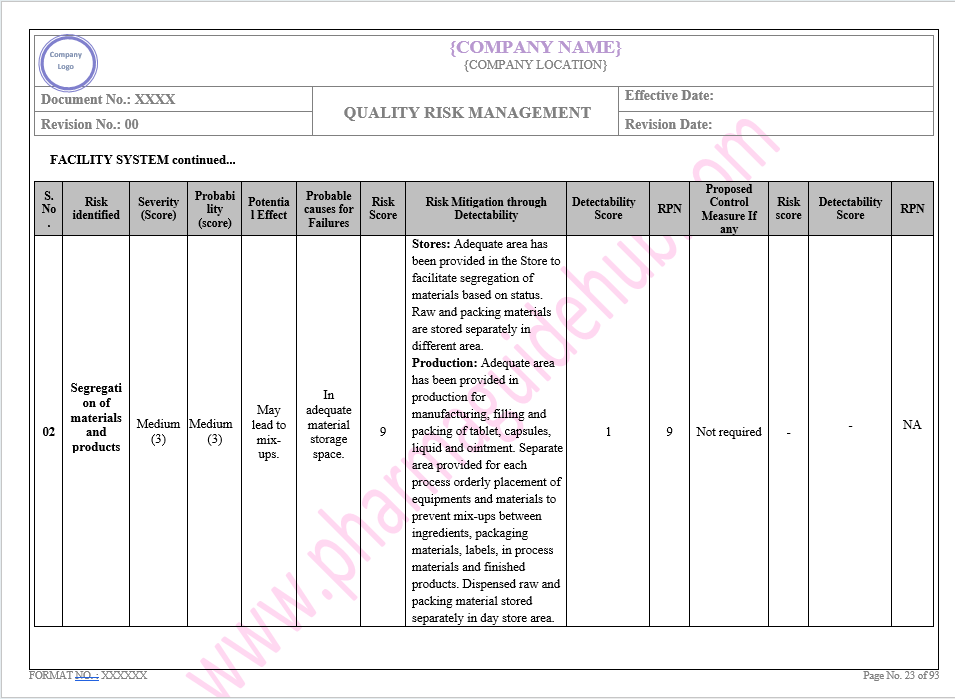
Key Parameter of Below Page:
Facility system
Find the below page for content
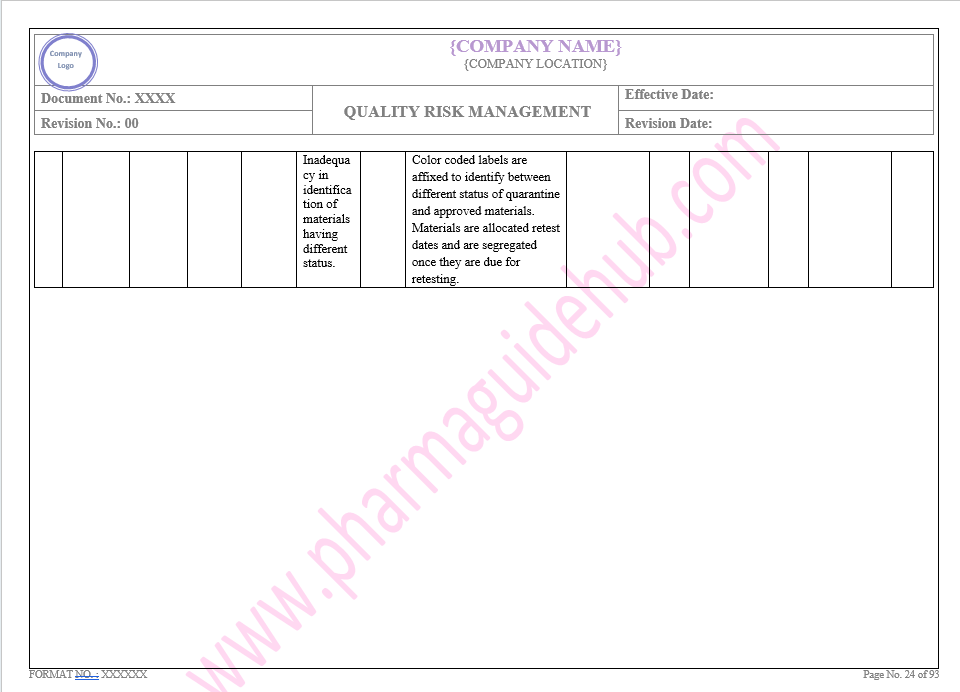
Key Parameter of Below Page:
Facility system
Find the below page for content
Click the link to download complete word file copy of this document:
https://pharmaguidehub.com/product/quality-risk-management/
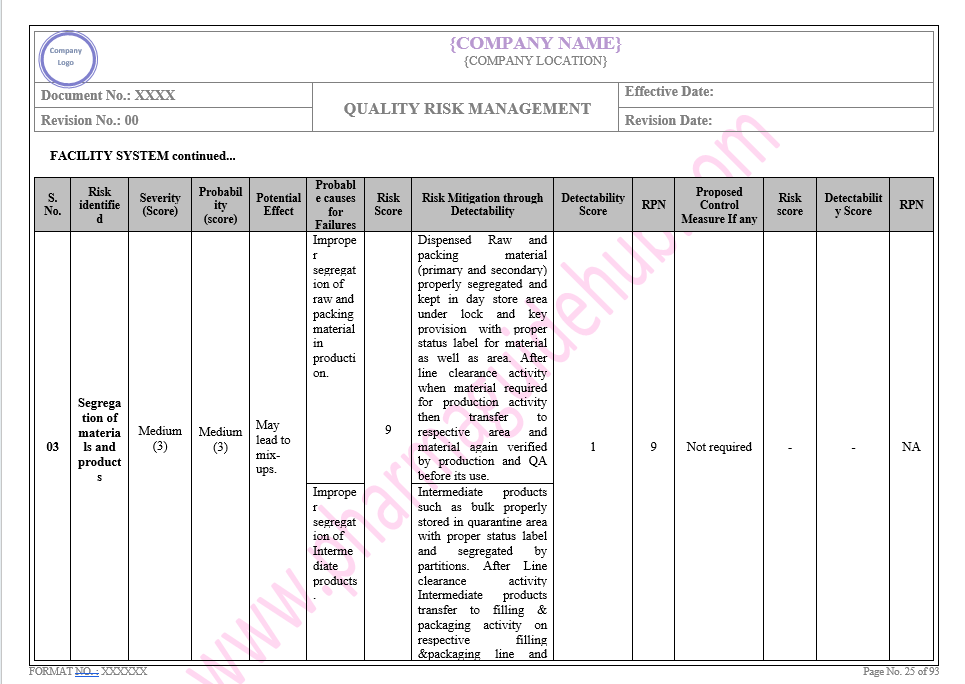
Key Parameter of Below Page:
Facility system
Find the below page for content
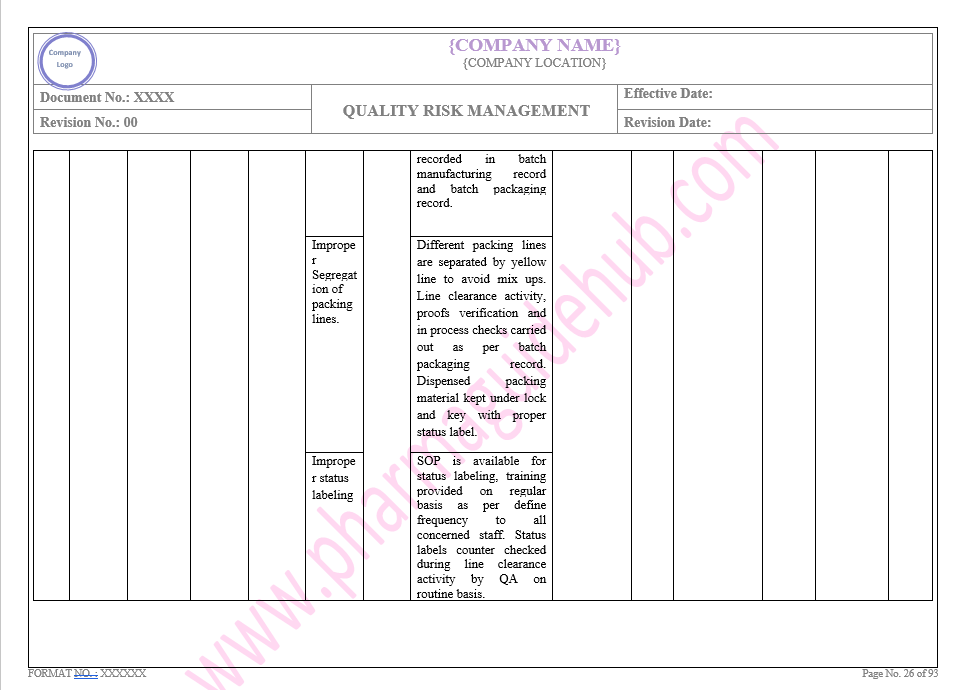
Key Parameter of Below Page:
Facility system
Find the below page for content
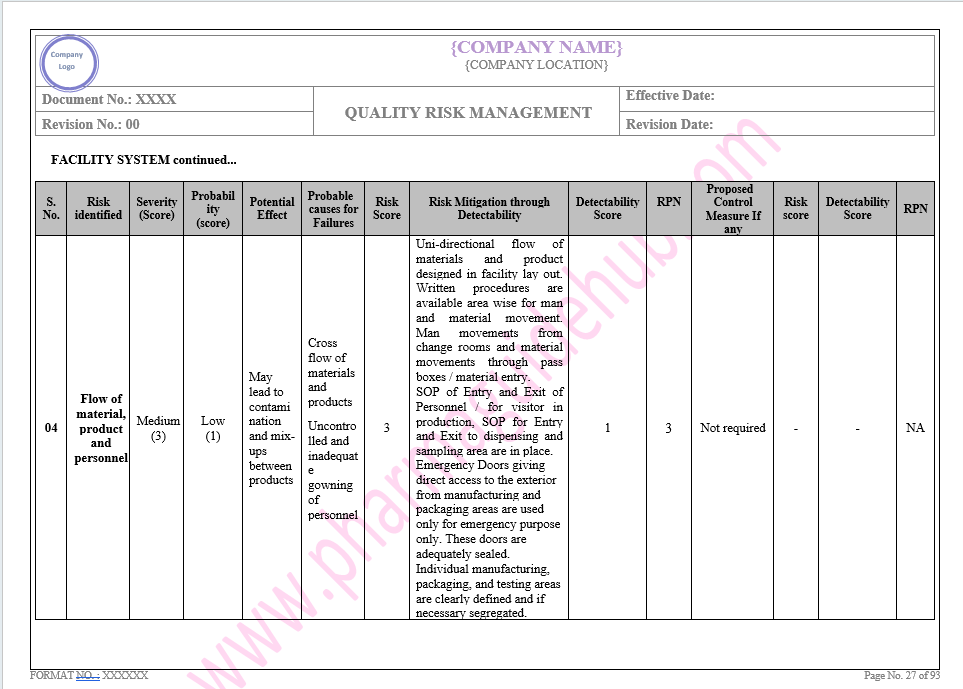
Key Parameter of Below Page:
Facility system
Find the below page for content
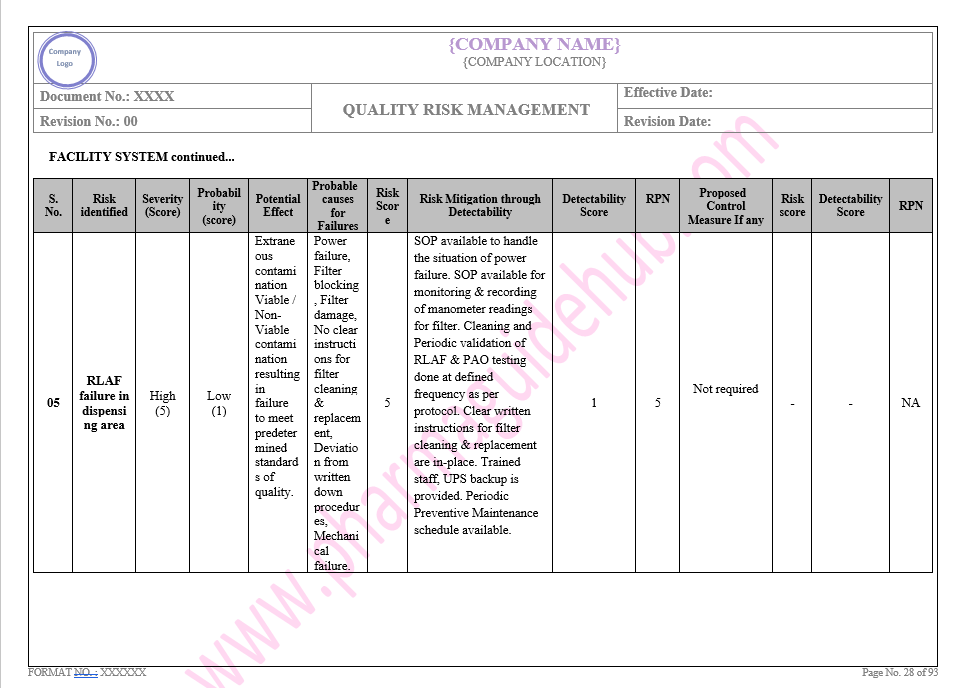
Key Parameter of Below Page:
Facility system
Find the below page for content
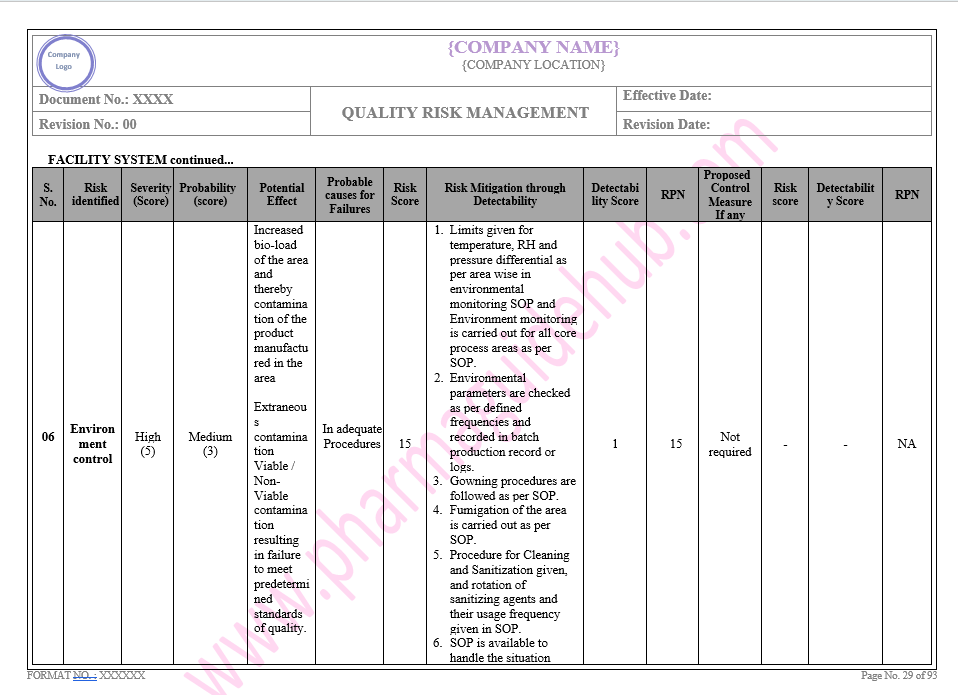
Key Parameter of Below Page:
Facility system
Find the below page for content
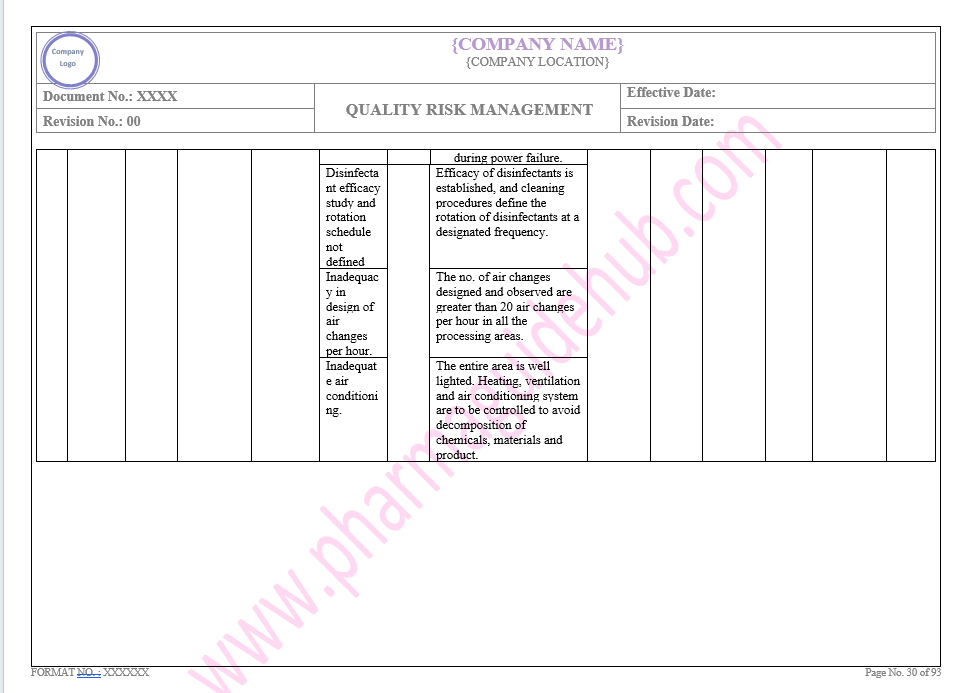
Key Parameter of Below Page:
Facility system
Find the below page for content
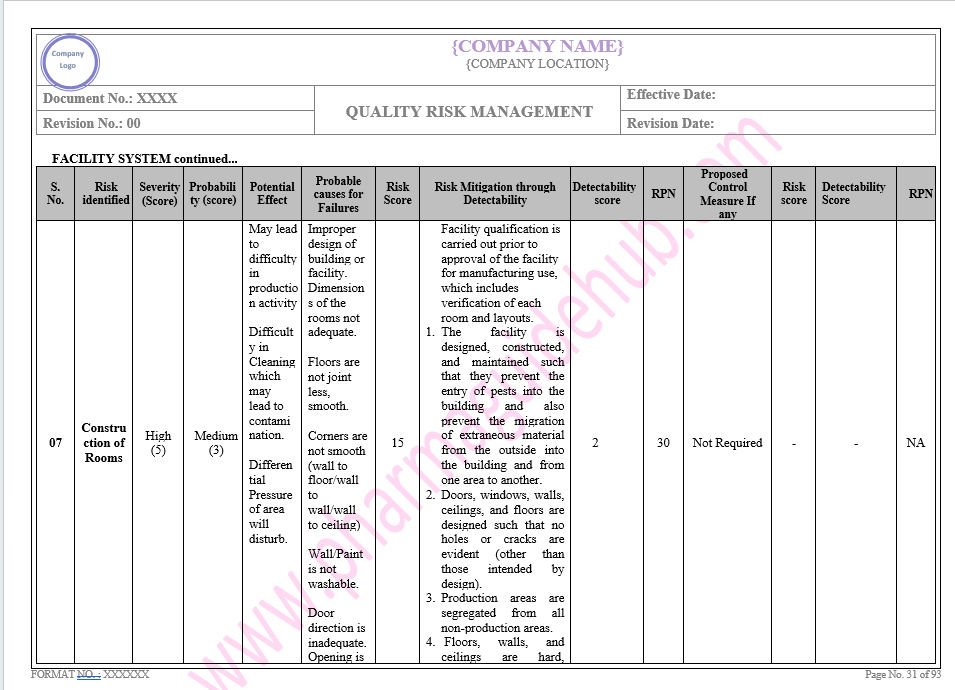
Click the link to download complete word file copy of this document:
https://pharmaguidehub.com/product/quality-risk-management/





