Objectives
Define sunscreens and the scientific of UV rays
Reasons for using sunscreens
Detail the various formulation of sunscreens available
Determination of the efficacy parameters
Regulatory requirements for sunscreens
Sunscreens
These are formulations applied onto the skin surface to protect against detrimental changes arising from the exposure to ultra violet rays.
Repeated exposure to sunlight causes sunburns, loss of elasticity and wrinkling of the skin.(photoaging)
Ultra violet rays have been associated with increased incidence of melanomas
Main cause of sunburns is UVB ,effects are acute and present with pain and erythema.
UVA causes delayed effects including wrinkling, elasticity loss and melanomas.
Ultraviolet Rays
They form part of the visible light.
UV spans of wavelength 200-400nm, UVC(180nm>), UVA(200-320nm), UVA( 320-400nm)
The UVC section of the band is not significant as the waves are filtered out by the ozone layer in the stratosphere.
UVB triggers production of melanin pigment and stimulates epidermal thickening resulting in a permanent skin tan.
UVA stimulates melanin already on the skin to produce short term tans. It however penetrates much deeper into the skin causing long term skin damage including mutations that precede cancers as well as skin aging.
The mutations result from DNA damage by free radicals produced when molecules on the skin surface are irradiated
Effects of UV rays
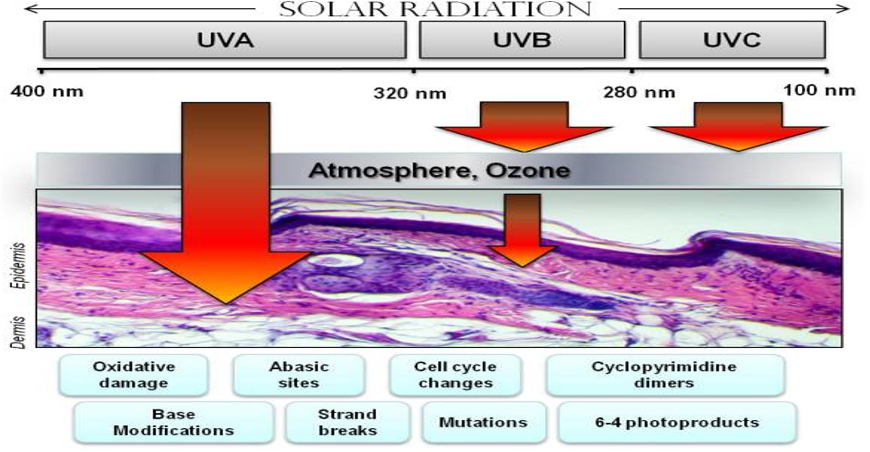
Observable effects of sunrays
Sun burns
Erythema
Loss of elasticity( loose or hanging skin)
Increased wrinkling
Melanomas in the long term
Determinants of extent of skin damage
Skin colour- Light skinned individuals are more susceptible to UV induced skin damage compared to those who are dark skinned .
Duration of exposure- The longer the exposure to sunlight the higher the magnitude of damage
Age– Children are more susceptible as their skins are undeveloped ( thin Stratum corneum)
Health status- Persons with existing skin disorders are more susceptible
Behavioral factors; Sun bathing, Tanning, Light clothing
History and modern use of sunscreens
Awareness of the damaging effects of the sunrays dates to prehistoric days. Egyptians, Greeks, chinese and Indians
Evidence of use include writings, wall carvings.
Modern use and production began in the early 1900’s coinciding with the scientific description of UV spectrum.
Earliest formulations were reported in USA(1928), PABA followed by Australia in 1934
More formulations became available after 1950s’
Classification
Based on mechanism of action
Chemical sunscreens: They absorb UV rays and converting it into heat energy that is reflected away from the skin.
Mineral sunscreens: They form an occlusive barrier that blocks, reflects or scatters the UV light
Classification based on mode of administration; Topical & Systemic
Sun protection factor: UVA, UVB, Broad spectrum
Organic sunscreens
These are organic chemical derived from natural sources and synthesis
They are further classified depending on the UV spectrum in which they afford the best protection.
UVB protectants;
PABA derivatives-
Cinnamates- Octisalate, Homosalate, Trolamine salicylate
Octocrylene
Ensulizole
Ethylhexyl triazone
UVA Filters
Benzophenones- e.g Oxybenzone, Sulibenzone,Dioxybenzone
Avobenzone
Meradimate
Bidisulizone disodium
Diethylaminohydroxyl benzoyl hexyhydrate
Ecanisole
Methylacralinate
Broad Spectrum sunscreen
They afford protection against both UVA and UVB
Ecamsule
Silatriazole
Bemotrizinol
Besotricozole

Inorganic Sunscreens
üMain constituents- Zinc oxide, Titanium oxide
Others
Talc
Iron oxide
Red veterinary petrolatum
Kaolin
Calamine
Ichthamol
Systemic sunscreens
These are taken orally and are absorbed systemically to afford protection. The act as antioxidants to scavenge radicals from the skin . Include
β- carotene, α- tocopherol, ascorbic acid, antimalarials( quinolines), Retinol, Selenium, green tea polyphenols, PABA, Antihistamines ,Aspirin, Indomethacin, Corticosteroids
Mechanism of action of sunscreens
Inorganic sunscreens
Afford skin protection by scattering and reflection of UV energy from the skin surface
Organic Sunscreens
Absorption of the UV energy by converting it to heat energy thus reducing its harmful effects and reduce the depth through which it can penetrate the skin.
Features of an ideal sunscreen
Chemical properties
Chemically inert
Non irritating
Photo stable
Compatible with other ingredients and formulation excipients
Soluble in appropriate solvents vehicles
Non odorous
Ideal features of sunscreen
Physical;
Non viscous to promote good spreadability
Aesthetically appealing to promote usage
Small particle size
Waterproof
Appropriate solubility in either aqueous or organic solvents
Ideal characteristics:
Functional attributes
Should afford protection against UV across a wider range of wavelength
Limited systemic absorption through the skin to minimize sensitization.
Others
Readily available
Inexpensive
Contaminant and microbe free
Other considerations
Convenience to the user
Patentability
Regulatory considerations
Special requirements for different target populations
Formulation
Sunscreens can be formulated as ;
Gels
Ointments
Lotions
Sprays
Sticks Powders
Traditionally Inorganic sunscreens were formulated as creams that were sticky and unpleasant.
Nanomization and micronization has allowed them to be formulated as pastes, emulsions and ointments
The aesthetic value is important to promote use.
Various sunscreens are usually combined in formulation to afford higher photo protection
They can also be included in other formulations such as makeup, bathing soap, hand sanitizers, and moisturizers
Suitable propellants are used in the case where the sunscreen is intended for use as a spray.(CFC, HC HCFC)
Emulsifying agents ( Eg.Agar, alginates, glycerol),
Other excipients
Surfactants
Buffering agents
Preservatives
Suspending agents
Moisturizers
Quality Control measures
Physical analysis. Includes organoleptic tests to check the appearance and feel of the product
Stability tests; Colour, phase separation and liquefaction. There should be no colour changes nor separation of phases in sunscreen formulations in the stability tests if they are to pass the quality tests. The absence of liquefaction provides strong evidence for the stability of the emulsions.
PH determination over time The PH value of sunscreen stored at different conditions is determined using a digital pH Meter. The pH tests are repeated for multiple emulsions or formulations after a defined period of storage
Determination of SPF in vitro using spectrophotometry
Level of water resistance for UVB: This test is conducted by immersion of a subject in a pool or spa for 40 minutes with a five minute rest in between(20-5-20). A sunscreen is considered to be water resistant if it retains no less than 50% of its SPF following immersion.
Microbiological stability For streptococcus aureus, psuedomonas aeroginosa, yeast and mould. Sunscreens like other topical formulations must be free from any microbial contamination that may render them deleterious to the users.
Measurement of photoprotection
Sun protection factor; refers to the ability of the sunscreen to prevent development of erythema upon exposure to UVB radiation
SPF = MED of protected skin/MED of unprotected skin (MED = minimal erythemal dose)
Application 2mg/cmsq
Grading
Low: SPF 2 – 15
Medium: SPF 15 – 30
High: SPF 30 – 50
Highest: SPF >50
Examples
Click the image to get sunscreen:

Persistent pigment darkening.
This measures establishes the ability of the sunscreen to protect against UVA light. The method is similar to that of establishing SPF Where the erythemic dose in a healthy subject covered with the sunscreen is compared to that obtained without using the sunscreen
Immune Protection Factor
Ability of sunscreen products to prevent UV-induced immunosuppression.
IPF is considered to correlate better with the UVA-protectiveness of a sunscreen than with its SPF
Clothing Factor
Fabric SPF is similar to SPF, except that fabric is used to protect the skin while testing, instead of a sunscreen.
Regulatory control
Regulation of sunscreen is variable across different jurisdictions.
In the USA, they are considered as OTC drugs
In European union and Japan sunscreens are considered to be cosmetics.
In Australia they are considered therapeutic goods and regulated as such. Regulation in
other countries in not well defined, most countries follow the USA or EU guidelines
Labelling requirements
Label must include
Manufacturers name and physical address
List of ingredients in order of predominance from the highest
Indication of the SPF
Optimal storage conditions
Cautionary information for patients who may be allergic to any of the ingredients
Challenges
Documented sensitivity to sunscreens
Some orgnanic sunscreens are absorbed through the skin resulting in systemic effects
Association with cancer induction especially when kaolin and talc is sourced from unverified sources( contamination with asbestos)
Inappropriate application methods that render the sunscreens ineffective
Click the image to get sunscreen:



