INVESTIGATION REPORT
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
1. Objective
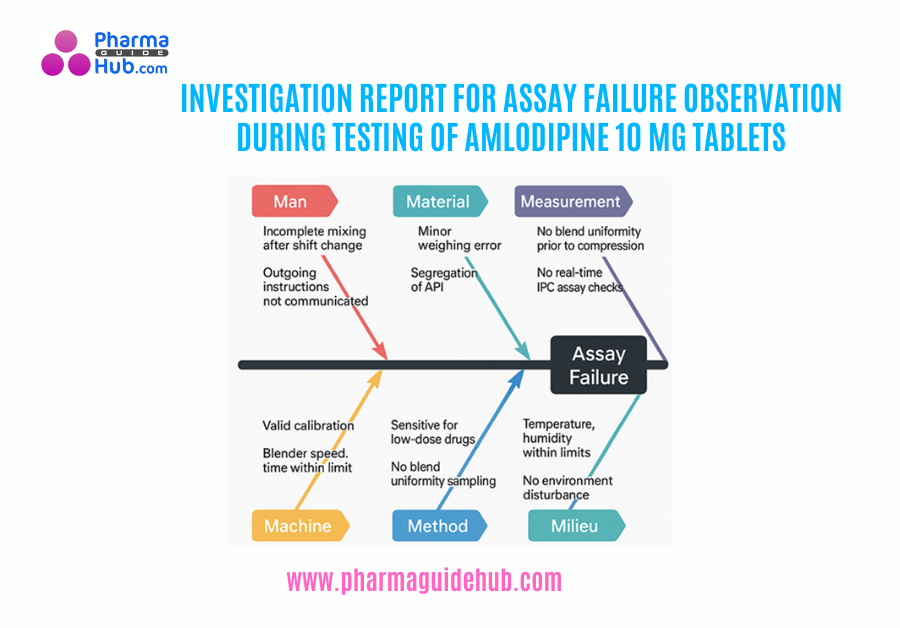
To investigate the root cause of assay failure observed during the analysis of Amlodipine 10 mg Tablet, batch number PGH/T/25-009, and to identify contributing factors at the manufacturing floor (Phase II), as Phase I laboratory-based investigation yielded satisfactory results.
Key Parameter of Below Page:
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
Find below pages for complete report

Key Parameter of Below Page:
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/investigation-report-for-assay-failure-observation-during-testing-of-amlodipine-10-mg-tablets/

Key Parameter of Below Page:
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
Find below pages for complete report

Key Parameter of Below Page:
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
Find below pages for complete report

Key Parameter of Below Page:
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
Find below pages for complete report

Key Parameter of Below Page:
Assay Failure Observation during Testing of Amlodipine 10 mg Tablets
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/investigation-report-for-assay-failure-observation-during-testing-of-amlodipine-10-mg-tablets/
YOU CAN ALSO READ:
INVESTIGATION REPORT FOR THE OBSERVATION OF BLACK PARTICLE IN COMPRESSED TABLET AFTER COMPRESSION
INVESTIGATION REPORT FOR WATER LEAKAGE FROM COMPRESSION ROOF
INVESTIGATION REPORT FOR LEAK TEST FAILURE OF BLISTER PACK
INVESTIGATION REPORT FOR WRONG BATCH CODING OBSERVED ON MONOCARTON DURING SECONDARY PACKING
INVESTIGATION REPORT FOR THE OBSERVATION OF A LIVE LIZARD IN THE TABLET QUARANTINE AREA
6M-Based Investigation on Tablet Chipping at Compression Stage
INVESTIGATION REPORT FOR RAW MATERIAL SPILLAGE DURING DISPENSING
INVESTIGATION REPORT FOR OBSERVATION OF FLY IN MANUFACTURING CORRIDOR
INVESTIGATION REPORT FOR LOWER YIELD AT COMPRESSION STAGE
STANDARD OPERATING PROCEDURE FOR THE HANDLING OF DEVIATIONS AND INCIDENTS





This was a really thorough report on the assay failure for Amlodipine 10mg tablets. It’s fascinating to see the level of detail involved in pharmaceutical quality control. I was curious, given the common use of Amlodipine for both hypertension and angina, if there’s any data suggesting that certain patient populations might be more susceptible to the specific issues leading to these assay discrepancies? I’m thinking particularly about how different patient responses could potentially complicate future investigations or even drug formulation in the long run. Thanks for such an insightful read!
Great detailed breakdown! It would be useful to explore how Phase II testing conditions might differ from Phase I, especially in terms of equipment or environmental factors that could impact the results.