Computer System Validation (CSV)
Computer System Validation (CSV) in the pharmaceutical industry is a critical process to ensure the reliability and accuracy of computerized systems used in drug development and manufacturing. It involves a series of activities to confirm that these systems meet predefined specifications and fulfill their intended purpose. CSV helps to maintain data integrity, protect patient safety, and comply with regulatory requirements.
Click the Link for IQ and OQ of computer system validation (CSV):
https://pharmaguidehub.com/computer-system-validation-csv-installation-operational-qualification-protocol-cum-report/
Performance Qualification (PQ)
Performance Qualification (PQ) in computer system validation for pharmaceuticals is crucial. It verifies that a system consistently performs according to user requirements and intended use in a simulated real-world environment. PQ involves testing the entire system, including hardware, software, and network components, to ensure it meets predefined specifications.
This process confirms the system’s reliability, accuracy, and ability to handle expected workloads, ensuring data integrity and compliance with regulatory standards like 21 CFR Part 11. Ultimately, successful PQ demonstrates that the computer system is fit for its intended use within pharmaceutical operations.

Key Parameter of Below Page:
Table of Content
Find below pages for complete protocol:

Key Parameter of Below Page:
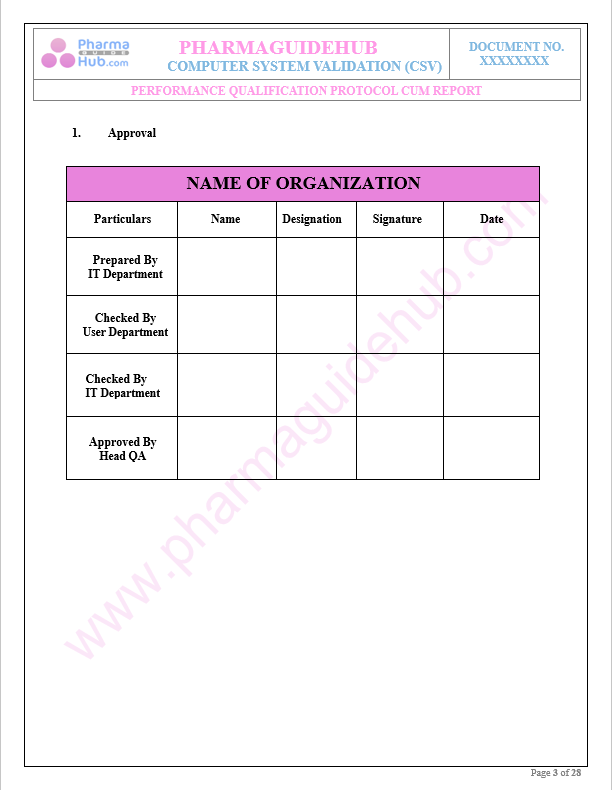
Approval
Find below pages for complete protocol:
Key Parameter of Below Page:
Purpose
Scope
Description
Find below pages for complete protocol:
Click the link to download word file copy of this document and Annexures:
https://pharmaguidehub.com/product/performance-qualification-pq-of-computer-system-validation-csv/

Key Parameter of Below Page:
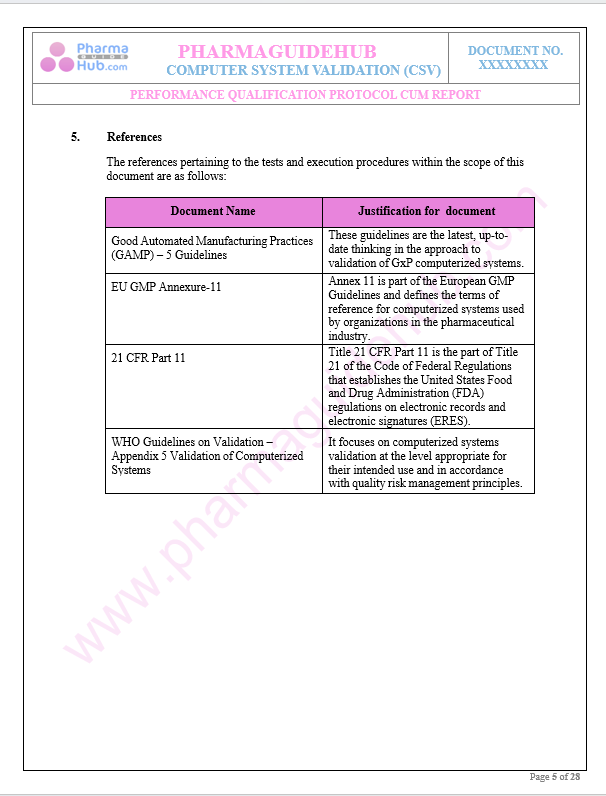
References
Find below pages for complete protocol:
Key Parameter of Below Page:
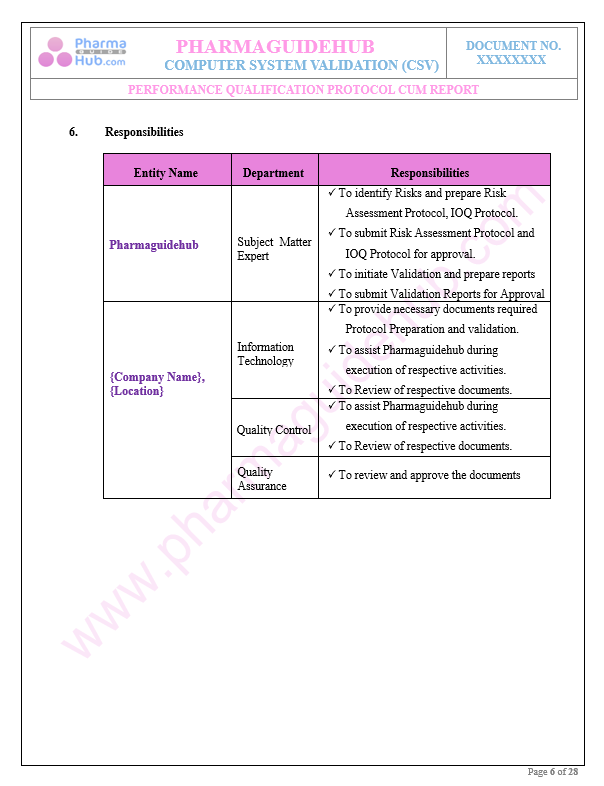
Responsibilities
Find below pages for complete protocol:
Key Parameter of Below Page:
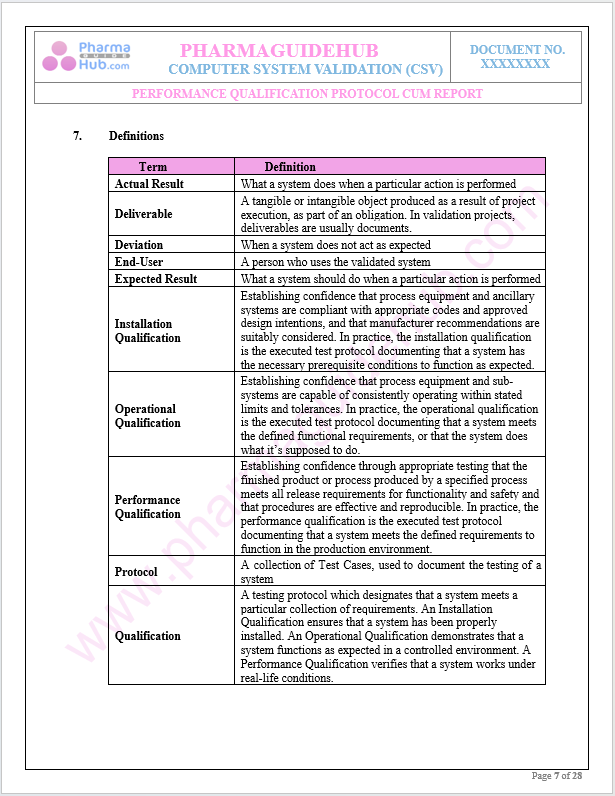
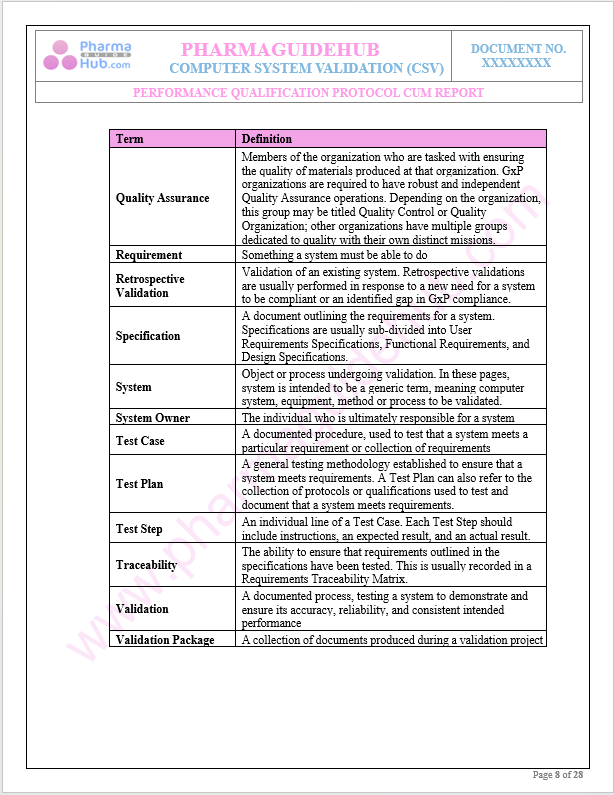
Definitions
Find below pages for complete protocol:
Key Parameter of Below Page:
Definitions
Find below pages for complete protocol:
Key Parameter of Below Page:
Execution Instructions
Find below pages for complete protocol:
Click the link to download word file copy of this document and Annexures:
https://pharmaguidehub.com/product/performance-qualification-pq-of-computer-system-validation-csv/

Key Parameter of Below Page:
Execution Instructions
Find below pages for complete protocol:
Click the to download the Performance Qualification of Computer system Validation (CSV) – Compiled Document
https://pharmaguidehub.com/product/performance-qualification-of-computer-system-validation-csv-compiled-document/

Key Parameter of Below Page:
Performance Qualification
Find below pages for complete protocol:

Key Parameter of Below Page:
Performance Qualification
Find below pages for complete protocol:
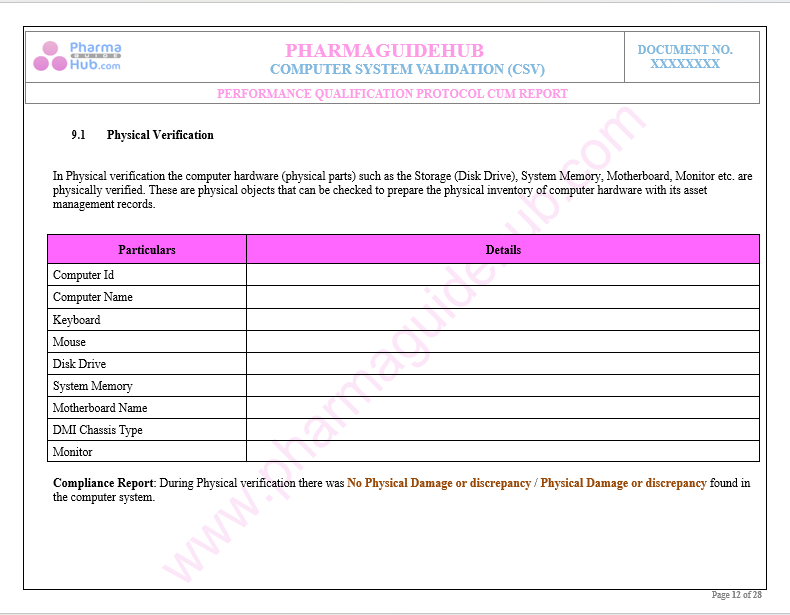
Key Parameter of Below Page:
Performance Qualification
Find below pages for complete protocol:
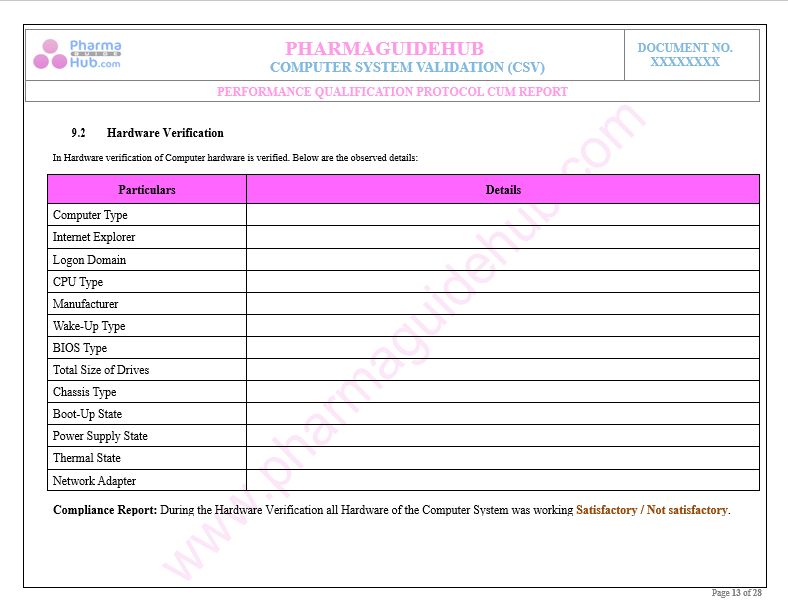
Key Parameter of Below Page:
Performance Qualification
Find below pages for complete protocol:
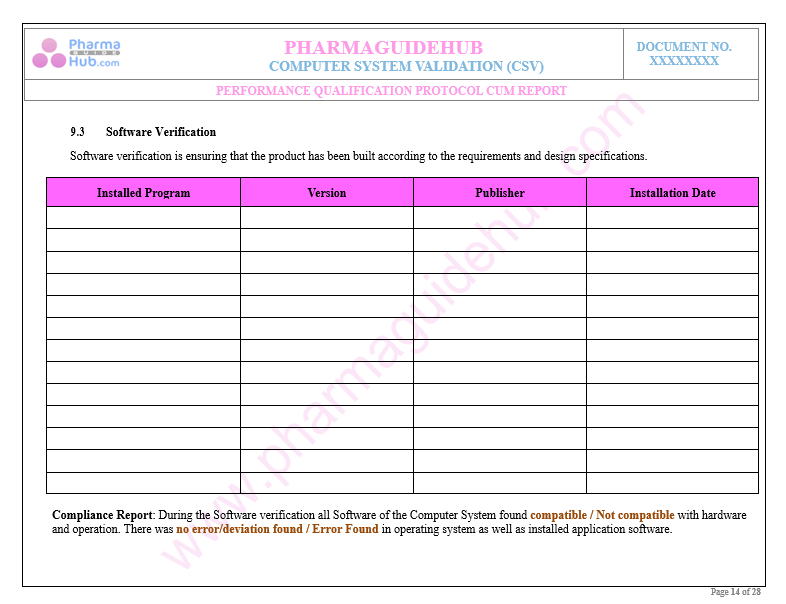
Key Parameter of Below Page:
Performance and Security Parameters Test Cases
Find below pages for complete protocol:

Key Parameter of Below Page:
Performance and Security Parameters Test Cases
Find below pages for complete protocol:
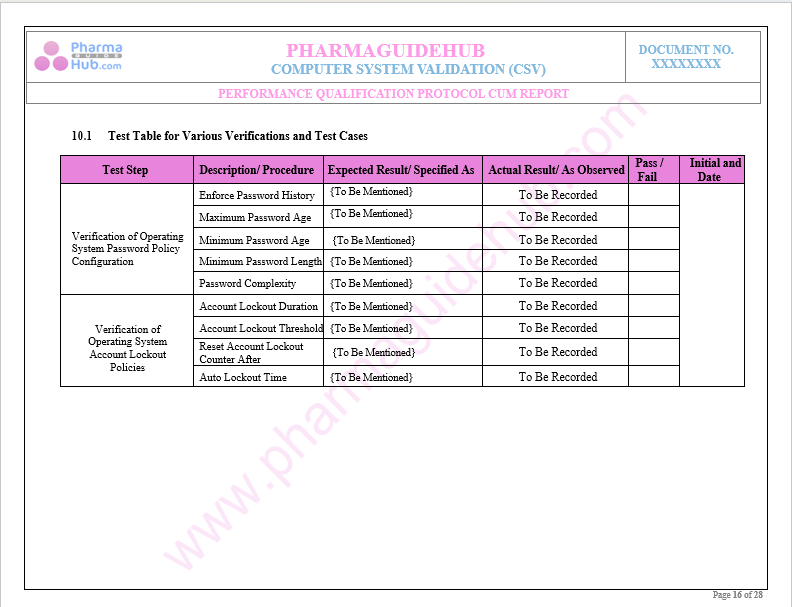
Key Parameter of Below Page:
Performance and Security Parameters Test Cases
Find below pages for complete protocol:
Click the link to download word file copy of this document and Annexures:
https://pharmaguidehub.com/product/performance-qualification-pq-of-computer-system-validation-csv/
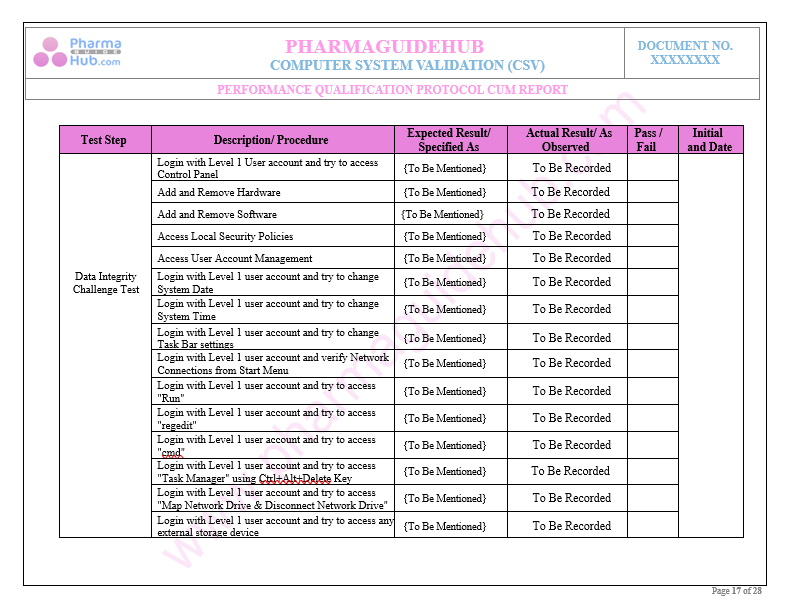
Key Parameter of Below Page:
Performance and Security Parameters Test Cases
Find below pages for complete protocol:
Click the to download the Performance Qualification of Computer system Validation (CSV) – Compiled Document
https://pharmaguidehub.com/product/performance-qualification-of-computer-system-validation-csv-compiled-document/
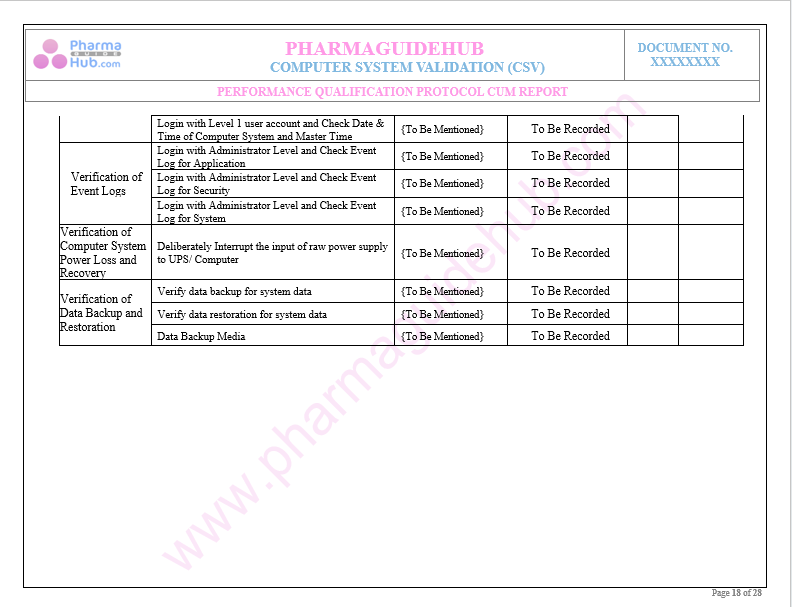
Key Parameter of Below Page:
Performance and Security Parameters Test Cases
Find below pages for complete protocol:
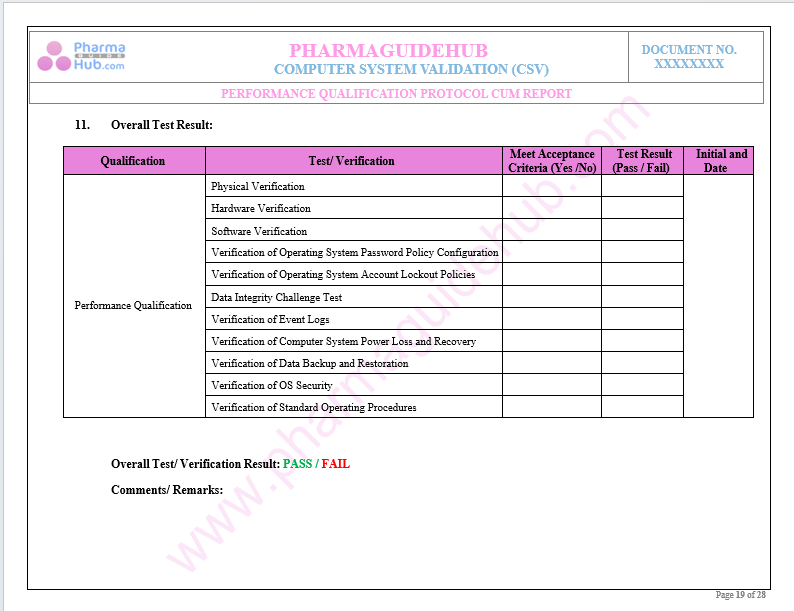
Key Parameter of Below Page:
Discrepancy Report
Find below pages for complete protocol:
Key Parameter of Below Page:
Discrepancy Report
Find below pages for complete protocol:
Key Parameter of Below Page:
Discrepancy Report
Find below pages for complete protocol:
Key Parameter of Below Page:
Discrepancy Report
Find below pages for complete protocol:
Click the to download the Performance Qualification of Computer system Validation (CSV) – Compiled Document
https://pharmaguidehub.com/product/performance-qualification-of-computer-system-validation-csv-compiled-document/
Key Parameter of Below Page:
Abbreviations
Find below pages for complete protocol:

Key Parameter of Below Page:
Abbreviations
Find below pages for complete protocol:

Key Parameter of Below Page:
Abbreviations
Benefit of Validation
Find below pages for complete protocol:
Click the link to download word file copy of this document and Annexures:
https://pharmaguidehub.com/product/performance-qualification-pq-of-computer-system-validation-csv/
Click the to download the Performance Qualification of Computer system Validation (CSV) – Compiled Document
https://pharmaguidehub.com/product/performance-qualification-of-computer-system-validation-csv-compiled-document/

Key Parameter of Below Page:
Benefit of Validation
Acceptance Criteria
Summary
Conclusion
Find below pages for complete protocol:

Key Parameter of Below Page:
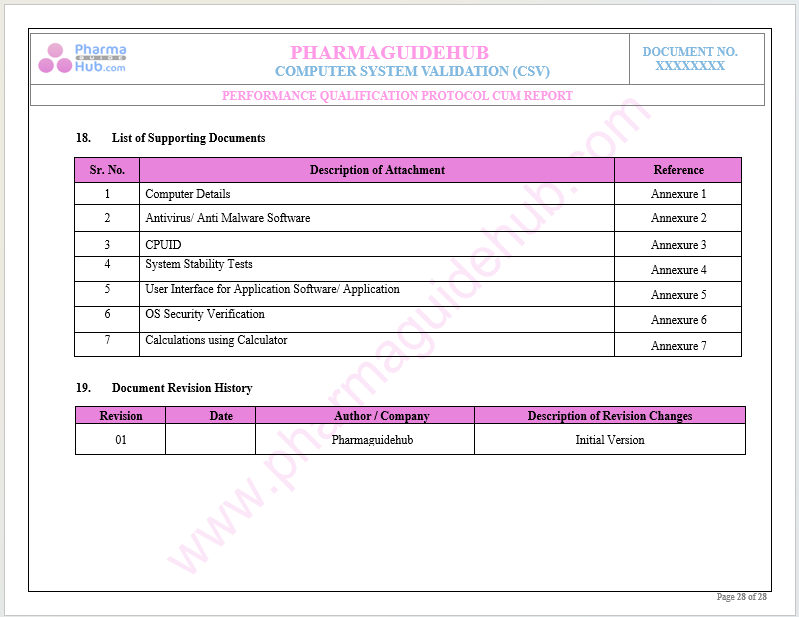
List of Supporting Documents
Document Revision History
Find below pages for complete protocol:
Click the link to download word file copy of this document and Annexures:
https://pharmaguidehub.com/product/performance-qualification-pq-of-computer-system-validation-csv/
Click the to download the Performance Qualification of Computer system Validation (CSV) – Compiled Document
https://pharmaguidehub.com/product/performance-qualification-of-computer-system-validation-csv-compiled-document/
You Can also Read:
COMPUTER SYSTEM VALIDATION (CSV)- INSTALLATION & OPERATIONAL QUALIFICATION PROTOCOL CUM REPORT
OPERATIONAL QUALIFICATION PROTOCOL CUM REPORT FOR FLUID BED PROCESSOR
INSTALLATION QUALIFICATION PROTOCOL CUM REPORT FOR FLUID BED PROCESSOR
INSTALLATION QUALIFICATION PROTOCOL CUM REPORT OF TABLET COATER FOR ACG PHARMA TECHNOLOGY
OPERATIONAL QUALIFICATION PROTOCOL CUM REPORT OF TABLET COATER FOR ACG PHARMA TECHNOLOGY
PERFORMANCE QUALIFICATION PROTOCOL CUM REPORT OF TABLET COATER FOR ACG PHARMA TECHNOLOGY
PERFORMANCE QUALIFICATION PROTOCOL CUM REPORT OF CHECK WEIGHER CW1200
ACTIVITY QUALIFICATION PROTOCOL FOR CALCULATOR
PERFORMACE QUALIFICATION PROTOCOL OF COLD CHAMBER
PERFORMANCE QUALIFICATION PROTOCOL FOR SEJONG 77 STATION DOUBLE ROTARY COMPRESSION MACHINE
PERFORMANCE QUALIFICATION PROTOCOL OF COMPRESSED AIR DISTRIBUTION SYSTEM






Asking questions are genuinely good thing if you are not understanding something entirely,
but this piece of writing provides nice understanding even.
I wanted to thank you for this great read!! I definitely enjoyed every little bit of it.
I have got you saved as a favorite to check out new things you post…
I couldn’t resist commenting. Very well written!