OBJECTIVE:
To lay down a Procedure for writing the Standard Operating procedure for transport validation
SCOPE:
This SOP is applicable for the transportation study of first three batches of all modes of route of transportation for specified container to know the transit conditions and its impact on product quality during transportation at {Company Name} {Location}.
RESPONSIBILITY:
Concerned department (Operating Executive/ Designee) shall be responsible for preparation of SOPs.
Operating Manager / Designee shall be responsible for review of SOPs.
QA Manager / Designee shall be responsible for Approval of SOPs.
ACCOUNTABILITY:
QA Manager Shall is accountable for implementation of SOPs.
PROCEDURE:
ABOUT TRANSPORT VALIDATION:
Transport validation in the pharmaceutical industry ensures that drugs and medical products maintain their quality, safety, and efficacy during transit. It involves documented testing to confirm that transportation methods, routes, and conditions—such as temperature, humidity, and vibration—meet regulatory requirements. Validation includes risk assessment, route mapping, data logging, and contingency planning. It is critical for products like vaccines and biologics, which are sensitive to environmental changes. Regulatory bodies like the FDA and EMA mandate strict compliance with Good Distribution Practices (GDP). Effective transport validation minimizes risks, protects public health, and supports product integrity throughout the supply chain.
Transportation validation Procedure:
Ensure proper working and initial adjustments of the calibrated data loggers before using them for study.
Ensure the Means of Transportation for proper storage environment for temperature.
The number of data loggers to be Implant shall be decided as per the route of transportation i.e. If Shipment is via Sea route two data loggers placed per transport storage container and if it’s Air Shipment One data logger to be placed per batch or as per customer suggestion.
Before dispatch of finished goods place the data loggers and record the location in Data logger Location sheet’ and verify the Transportation Checklist (annexure I).
Place the data loggers into separate corrugated box and label the corrugated boxes with details like “DATA LOGGER KEPT INSIDE” and “DATA LOGGER ARRANGED BY AND CHECKED BY, and also tape that box with red.
Arrange these boxes on pallets in such a way that these should be clearly visible (i.e., in front of pallets).
On receipt of consignment responsible person verify the container for any physical damage. If any damage is observed inform to the manufacturer and record the Date and time of receiving Consignment. The concerned person will remove the data logger from the corrugated box and generate the data in PDF.
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/transport-validation/
PRODUCT DETAILS:
Product Name, Dosage Form, Batch No., Distribution quantity, Primary pack details/pack configuration
Requirement for validation study:
Ensure the packing materials shall be available as per the approved SOP / Specification.
Ensure the availability of calibrated data loggers for the temperature mapping. Configuration (Recording interval) of the data logger shall be done in a computer using dedicated software as per the temperature mapping requirement/one time used data logger.
A pre-transport validation study shall be performed with the simulated product and packaging materials i.e. without actual drug product with minimum 10 data loggers or as per customer suggestion shall be placed for the validation study.
Whole transportation procedure shall be followed from loading destination (to first unloading destination (air port).
A conclusion of the data obtained from pre-transportation validation study shall be made and report shall be prepared with final recommendation.
Three commercial consignments shall be considered for the transport validation study. In each consignment minimum 10 data loggers or as Per Customer Suggestion shall be placed for the validation study.
The details of the vehicle used for the transport validation study shall be described in the performance qualification report. Packing/Labeling of the drug product shall be done as per approved procedure.
Calibrated data logger shall be placed inside the secondary package. Ensure the recording of the data logger shall be in ON condition (Press start button).
The details of the data logger used for the validation study shall be described in the performance qualification report.
Package containing the data logger shall be labeled for easy identification.
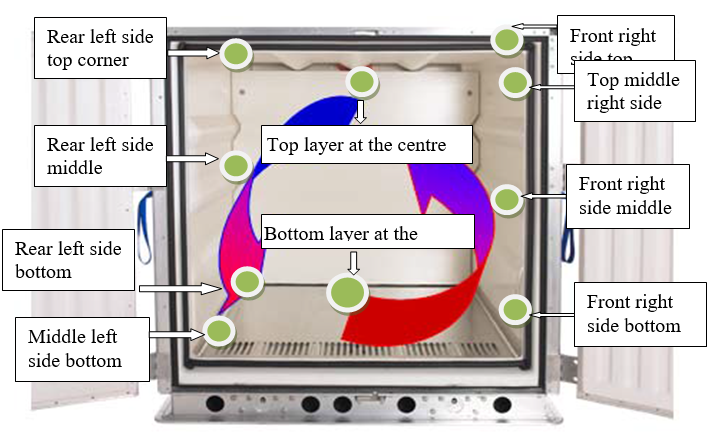
Data loggers to be placed in such a manner to represent whole of the geometry of the vehicle, diagonally opposite to each other on top, middle and bottom corners of the container or as per customer suggestion. Schematic representation / photograph of the location (Placement) of the data logger shall be represented in the validation report.
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/transport-validation/
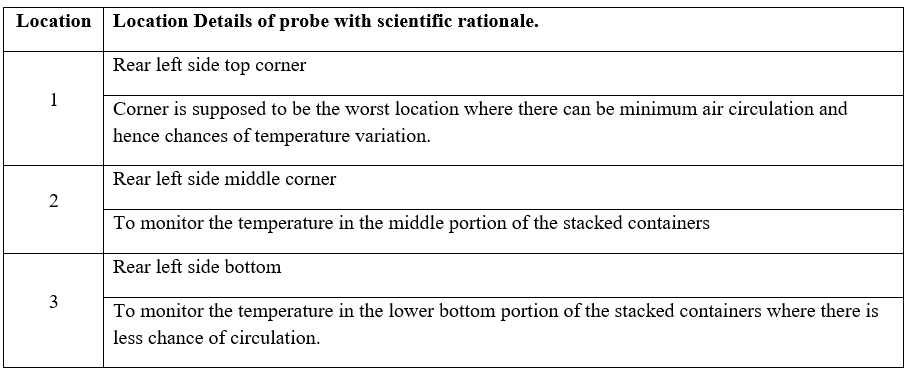
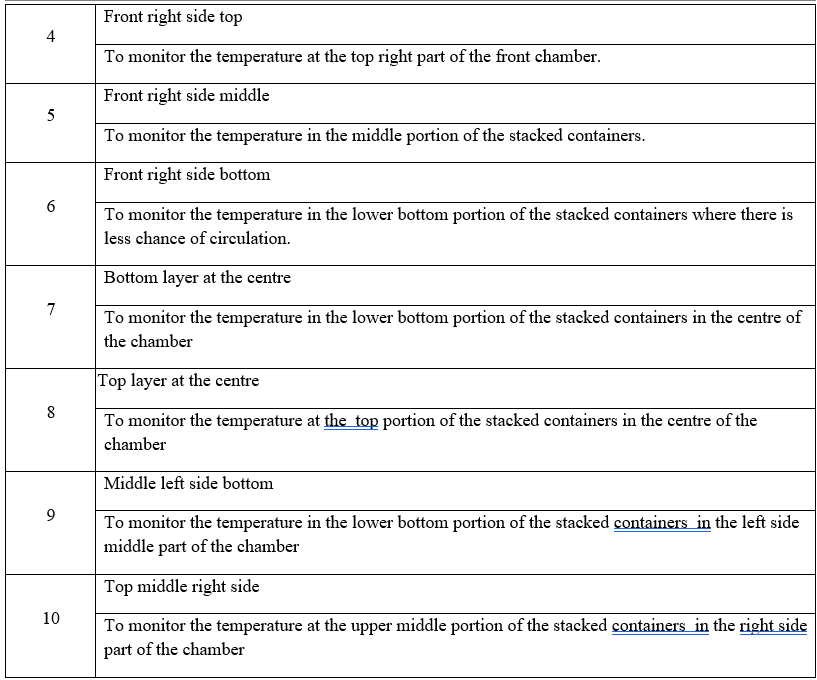
Ensure before transportation there is no physical defect or deformation of the packed material.
Nutraceutical products manufactured shall be dispatched after packaging as per the packing system to the customer location via road/air/sea route employing the designated transportation service.
At the receiver end, these recorded data loggers shall be taken from the identified packaging containers and data shall be downloaded by the sent consignee. Temperature monitored data shall be sent back through courier to product manufacturer /customer.
The data logger during the transportation shall be downloaded in the computer & printout shall be taken & reviewed for compliance as per the acceptance criteria.
Temperature mapping data of data logger during transportation shall be recorded in Annexure-II.
STORAGE CONDITION:
Temperature condition of storage and distribution NMT 25°C or as per product requirement.
TEMPERATURE MONITORING EQUIPMENT AND DEVICES:
Calibration Status of measuring devices shall be recorded in transport validation report.
A calibrated, external data logger capable of displaying and recording temperature shall be used for recording the temperature readings during the transport validation study.
The data loggers shall have an in-built real time clock to record temperature at predetermined time intervals.
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/transport-validation/
PROTOCOL CUM REPORT CONTENTS FOR TRANSPORT VALIDATION:
Protocol cum report pre approval
Objective
Scope
Responsibility
Reason for validation
Site of study
Pre-requisites for transport validation
Product information
Transport validation methodology
Procedure
Evaluation
Acceptance criteria
Procedure of start/stop of data logger
Deviation (if any
Summary
Conclusion
Recommendation
List of attachments
Abbreviations
Protocol cum report post approval
Revision history
REFERANCE:
In House
ANNEXURES:
| ANNEXURE No. | TITLE OF ANNEXURE |
| Annexure-I | Checklist for transport validation procedure |
| Annexure-II | Transport validation protocol cum report |
ENCLOSURES: SOP Training Record.
DISTRIBUTION:
| Controlled Copy No. 01Controlled Copy No. 02 | : : | Manager Quality Assurance Warehouse Department |
| Master Copy | : | Quality Assurance Department |
ABBREVIATIONS:
| SOP | : | Standard Operating Procedure |
| No. | : | Number |
| QA | : | Quality Assurance |
| % | : | Percentage |
REVISION HISTORY:
CHANGE HISTORY LOG
| Revision No. | Details of Changes | Reason for Change | Effective Date |
| 00 | New SOP | Not Applicable | To Be Written Manual |
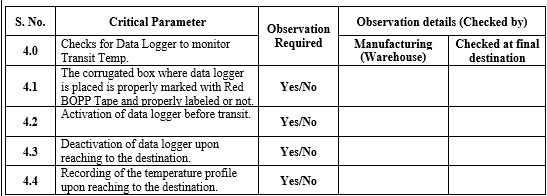
ANNEXURE- I
CHECKLIST FOR TRANPORT VALIDATION PROCEDURE



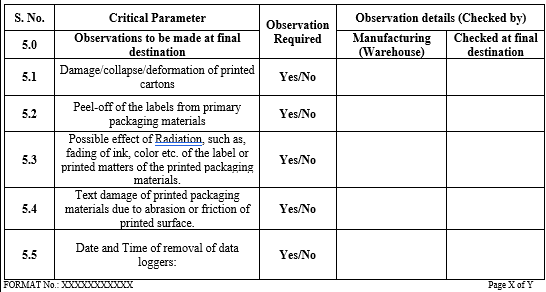
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/transport-validation/
ANNEXURE-II
TRANSPORT VALIDATION PROTOCOL CUM REPORT

Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/transport-validation/
You Can Also Read:
STANDARD OPERATING PROCEDURE FOR INVESTIGATION IN PHARMACEUTICAL INDUSTRY
INVESTIGATION OF OUT OF SPECIFICATION (OOS)
STANDARD OPERATING PROCEDURE FOR THE HANDLING OF DEVIATIONS AND INCIDENTS
COMPUTER SYSTEM VALIDATION (CSV)PERFORMANCE QUALIFICATION PROTOCOL CUM REPORT (PQ)





