Hold time – The established time period for which dispensed raw materials, intermediates (in-process materials), and bulk dosage form awaiting final packing may be held under specified conditions and will remain within the defined specifications. Intermediate (in-process materials): Partly processed product that must undergo further manufacturing steps before it becomes a bulk product. Hold time can be considered as the established time period for which materials (dispensed raw materials, intermediates, and bulk dosage form awaiting final packaging) may be held under specified conditions and will remain within the defined specifications.
Hold-time studies establish the time limits for holding the materials at different stages of production to ensure that the quality of the product does not produce results outside the acceptance criteria during the hold time.
The present study involves the determination of hold time through physico-chemical and microbiological analysis of intermediate/semi-finished products.
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES :
Find below pages for complete report

Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/
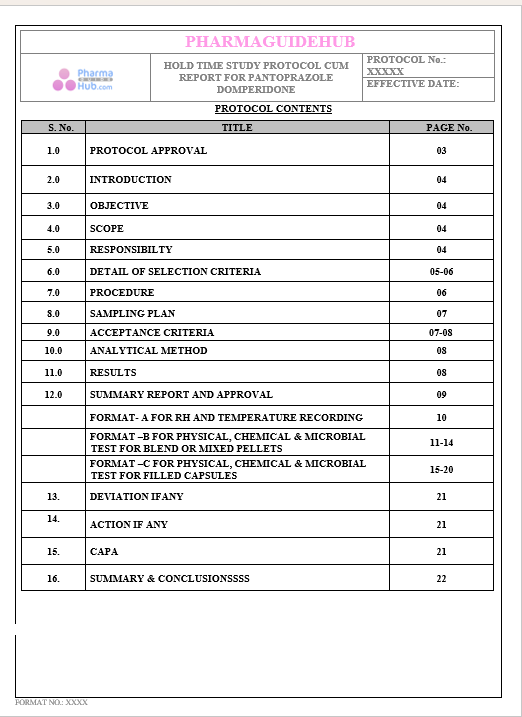
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:

Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/
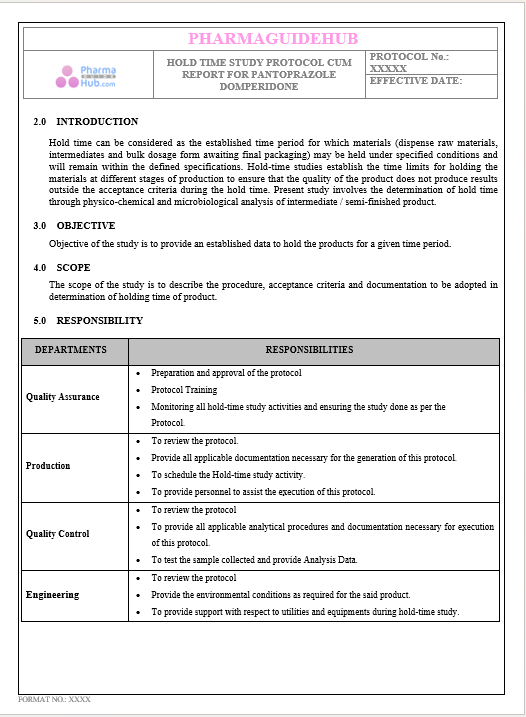
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/

Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/

Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/
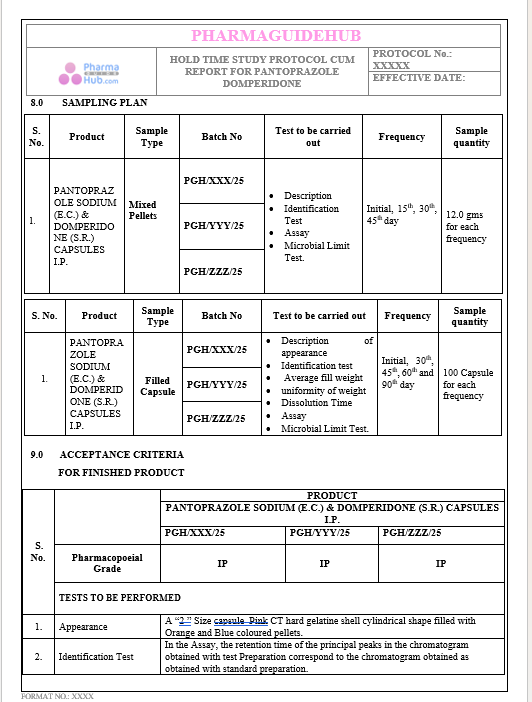
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
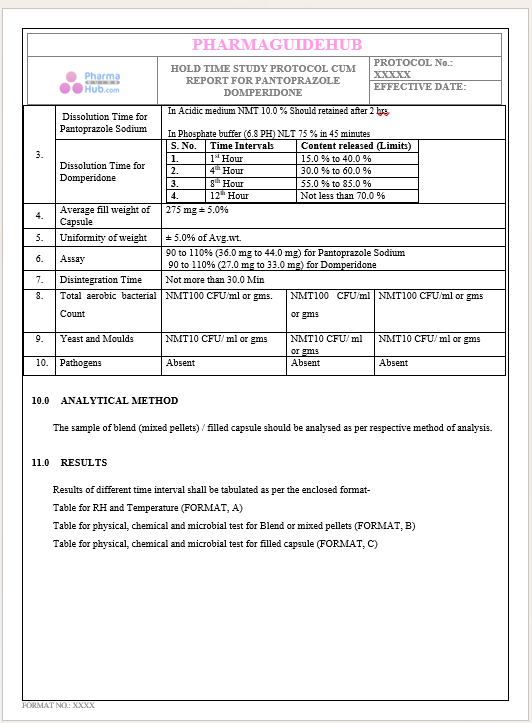
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
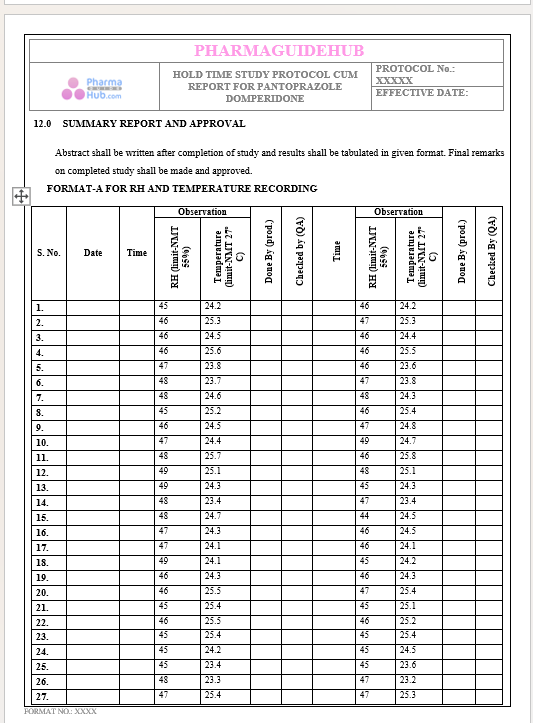
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/
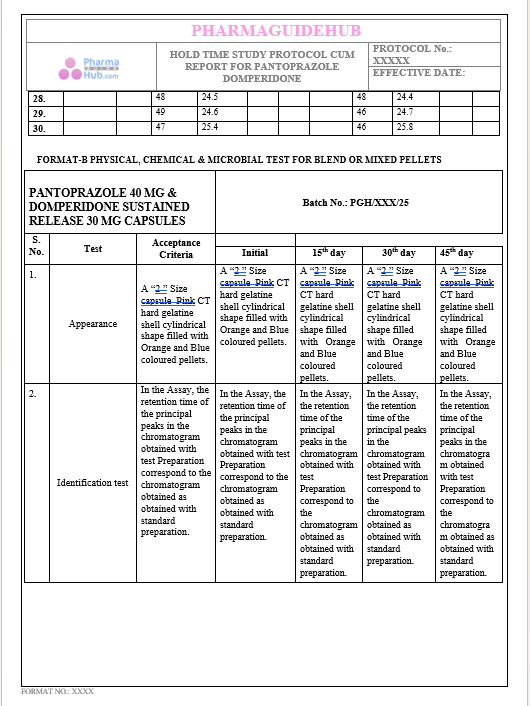
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
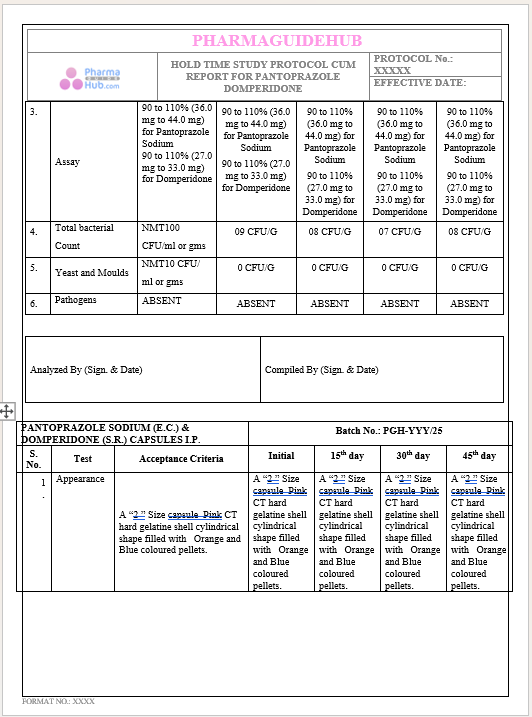
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
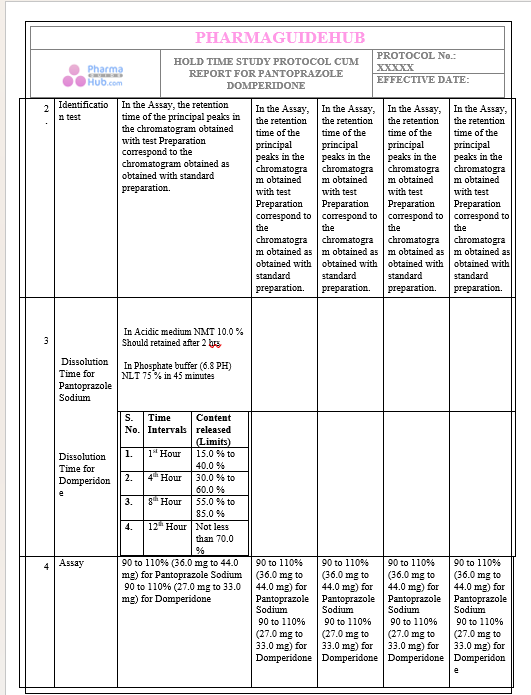
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
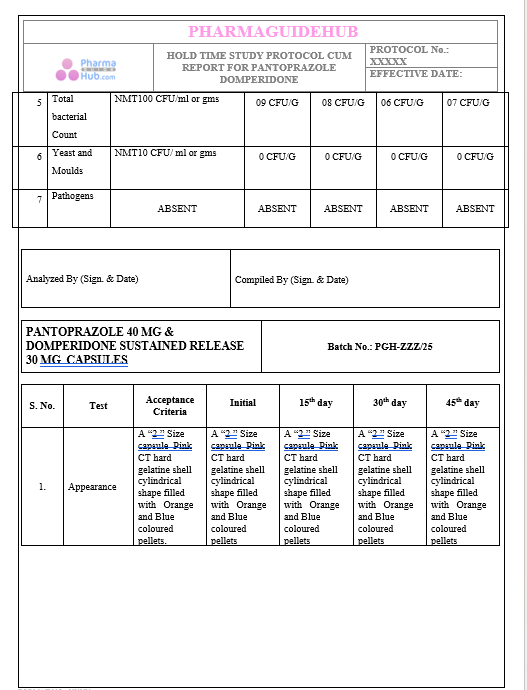
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
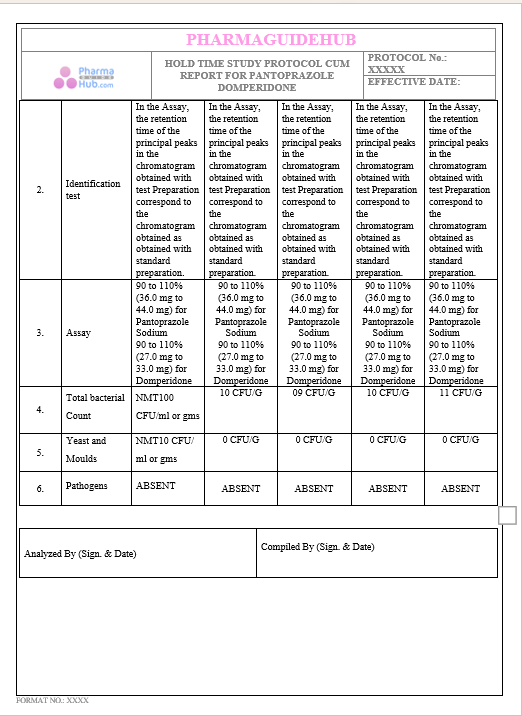
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/
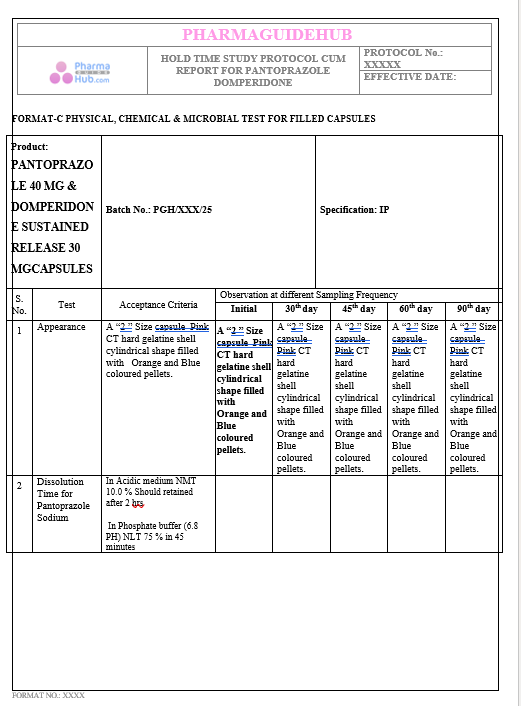
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
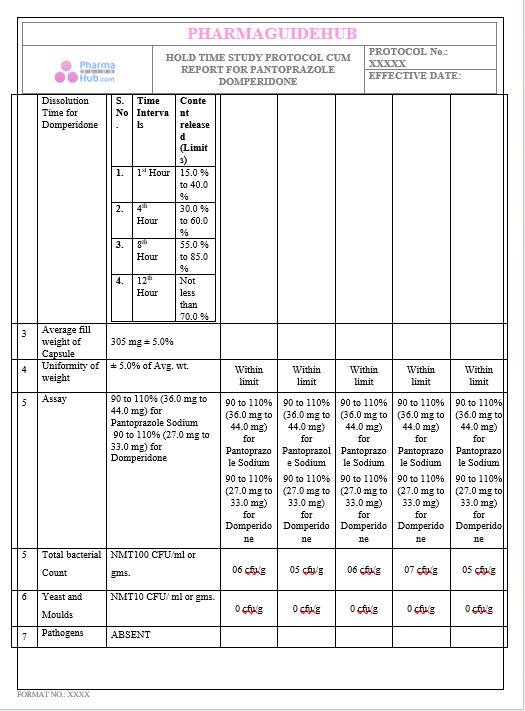
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
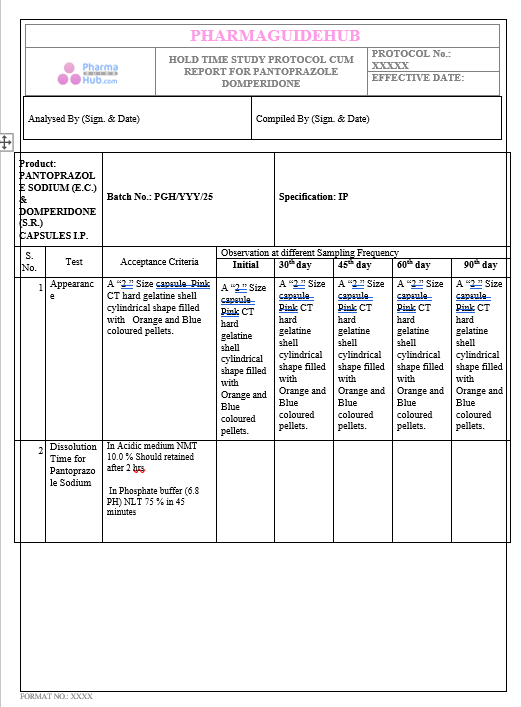
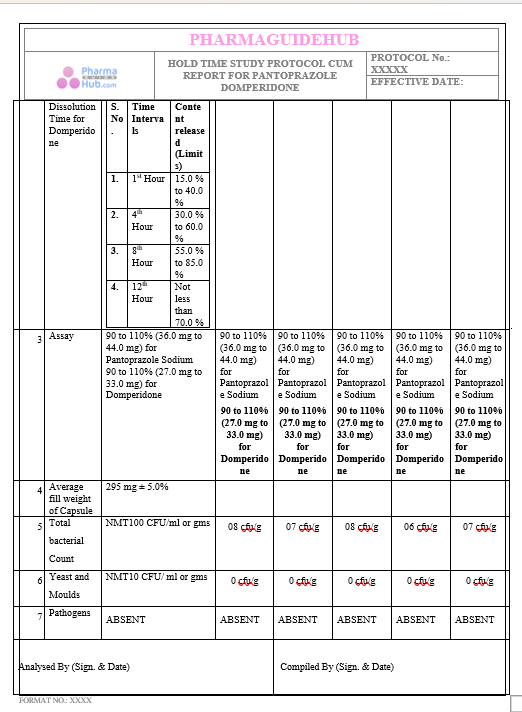
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/
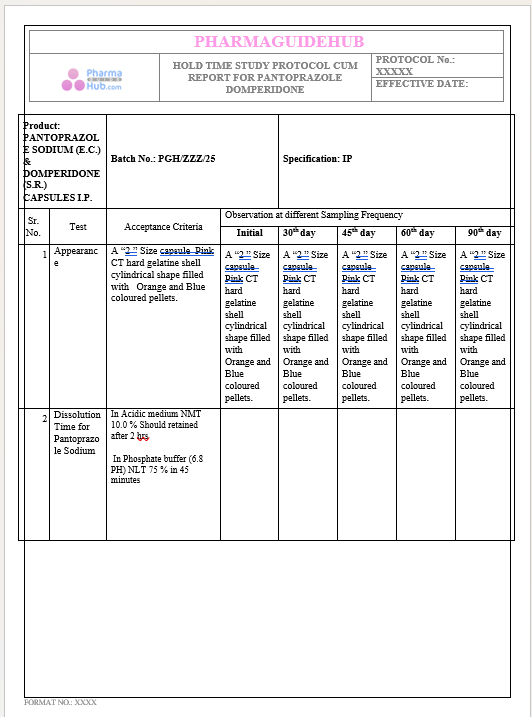
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
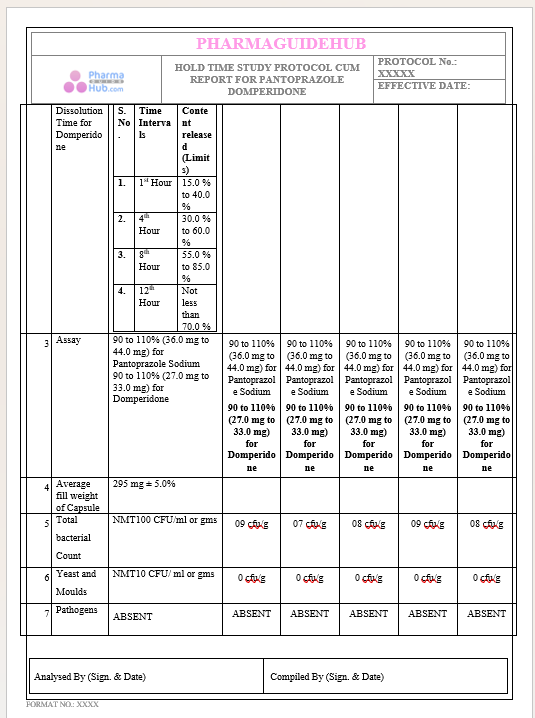
Key Parameter of the Below Page:
HOLD TIME STUDY PROTOCOL CUM REPORT FOR PANTOPRAZOLE DOMPERIDONE CAPSULES:
Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/

Click the link to download a word file copy of this document:
https://pharmaguidehub.com/product/hold-time-study-protocol-cum-report-for-pantoprazole-domperidone/





