During the WHO GMP inspection of a pharmaceutical plant, Name AABBCC, an observation was made that CAPA SYSTEM WAS INEFFECTIVE, AND REPEAT DEVIATIONS WERE OBSERVED. This has been received.
The compliance report along with supporting documents is to be submitted within 30 days.
1. Context of Observation
During the WHO GMP inspection at AABBCC pharmaceutical manufacturing facility, it was observed that the Corrective and Preventive Action (CAPA) system was not functioning effectively, resulting in recurrence of similar deviations.
Although a written SOP for CAPA management was available and implemented within the Pharmaceutical Quality System (PQS), the following gaps were identified:
- Repeat deviations were observed for similar root causes.
- CAPA effectiveness verification was inadequate.
- Trend analysis of recurring deviations was not effectively performed.
- CAPA closure was based primarily on documentation completion rather than objective effectiveness assessment.
- No robust mechanism was available to identify recurrence trends and systemic failures.
- Inadequate escalation of repeat deviations to management review.
This observation indicates weakness in the effectiveness of the CAPA system and continuous improvement process.
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-capa-system-was-ineffective-and-repeat-deviations-were-observed/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
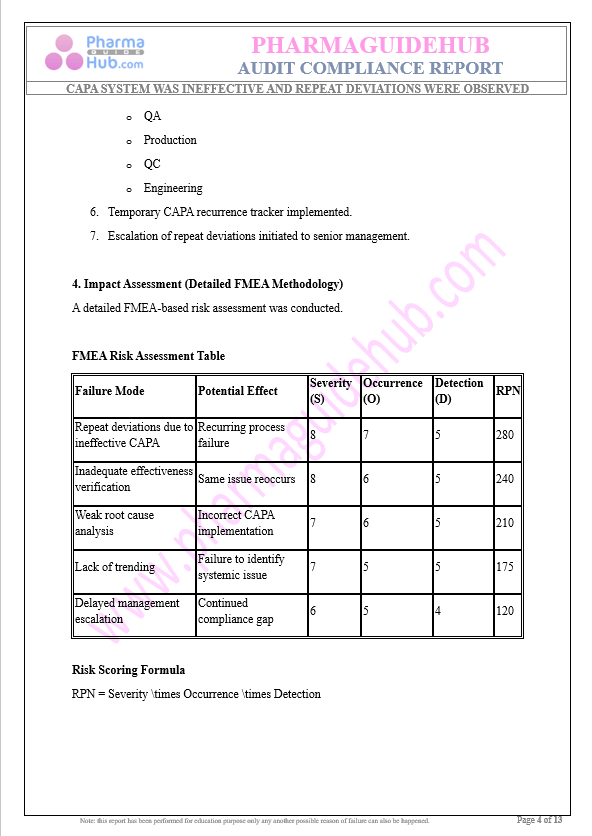
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-capa-system-was-ineffective-and-repeat-deviations-were-observed/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
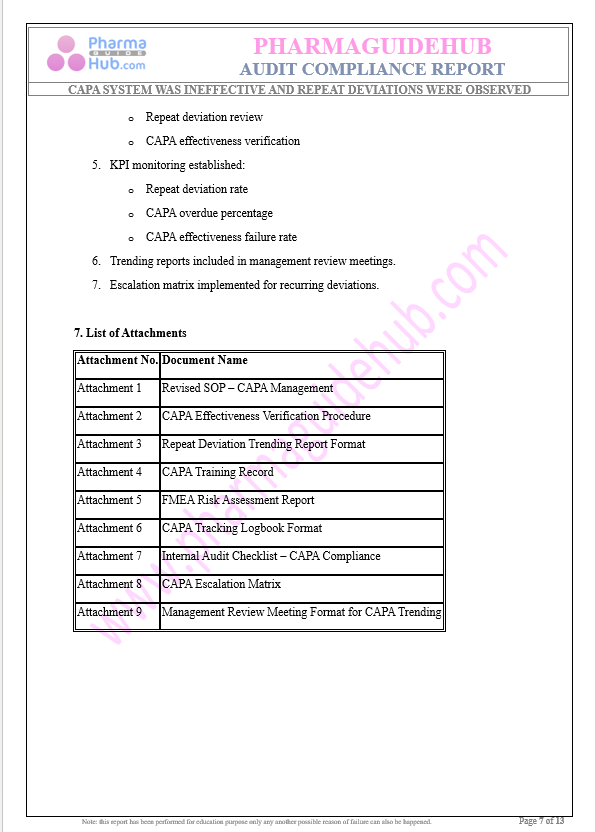
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
SOP FOR CORRECTIVE AND PREVENTIVE ACTION (CAPA) IN PHARMACEUTICAL

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
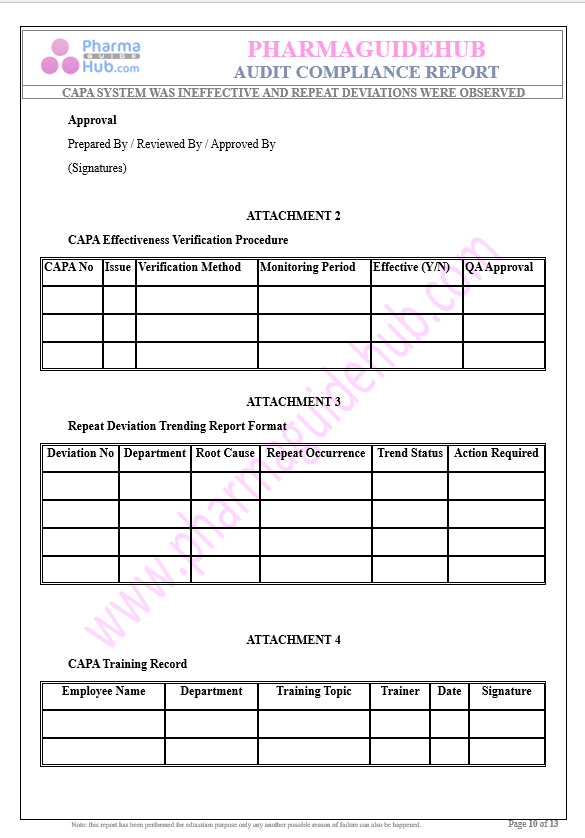
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-capa-system-was-ineffective-and-repeat-deviations-were-observed/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-capa-system-was-ineffective-and-repeat-deviations-were-observed/





