During the WHO GMP inspection of a pharmaceutical plant, Name AABBCC, an observation was made that NO PERIODIC QUALITY RISK MANAGEMENT (QRM) EXERCISES WERE PERFORMED. This has been received.
The compliance report along with supporting documents is to be submitted within 30 days.
Context of Observation
During the WHO GMP inspection of the pharmaceutical manufacturing facility AABBCC, the following observation was raised:
“No periodic Quality Risk Management (QRM) exercises performed.”
Although an approved SOP for Quality Management Review and provision for conducting Quality Risk Management (QRM) activities exists within the quality system, it was observed that:
- Periodic QRM exercises were not performed as per defined frequency.
- Risk review records were not available for ongoing processes/products.
- Periodic risk evaluation was not integrated into Management Review activities.
- No documented evidence demonstrating trend-based risk reassessment was available.
This observation indicates inadequate implementation and monitoring of the QRM system despite procedural availability.
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-no-periodic-quality-risk-management-qrm-exercises-performed/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
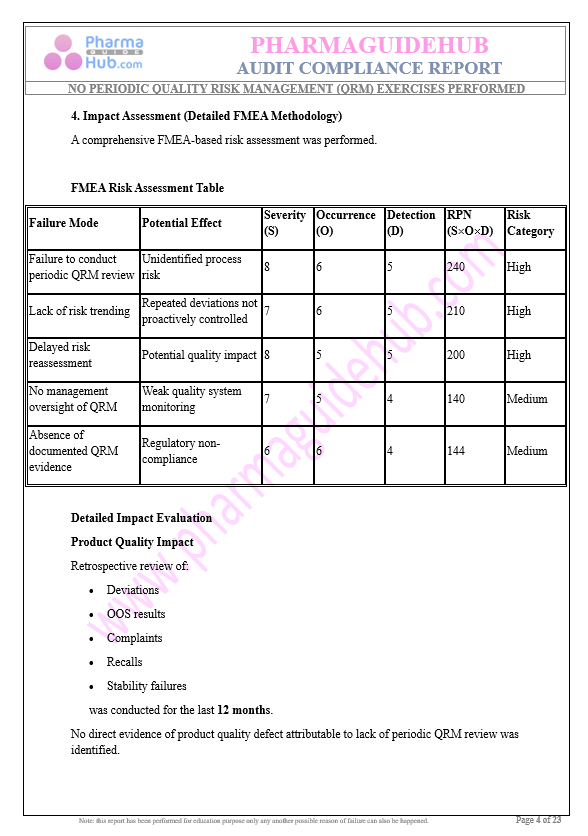
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-no-periodic-quality-risk-management-qrm-exercises-performed/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-no-periodic-quality-risk-management-qrm-exercises-performed/
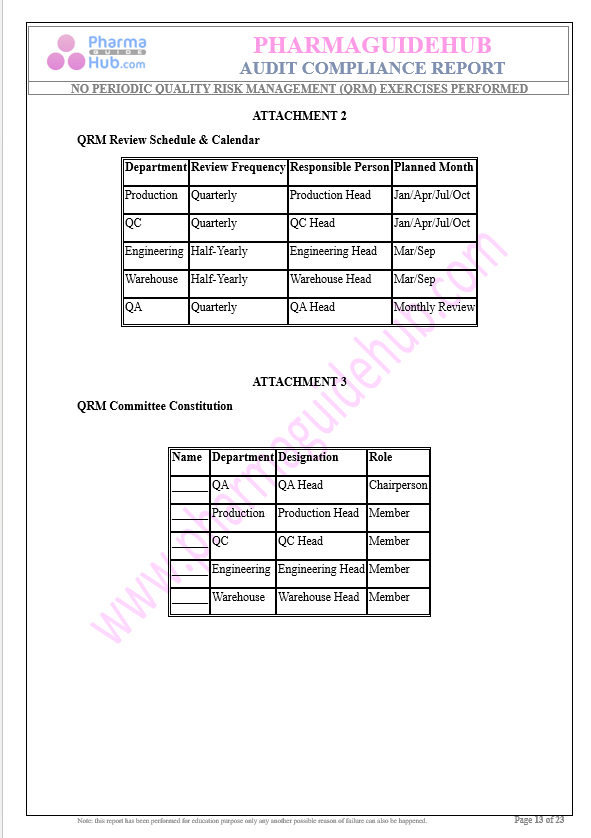
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
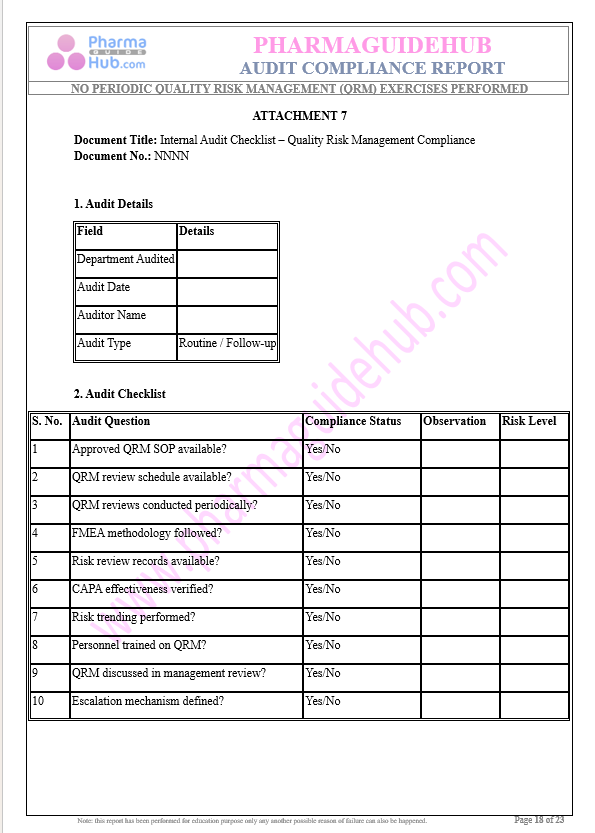
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-no-periodic-quality-risk-management-qrm-exercises-performed/

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
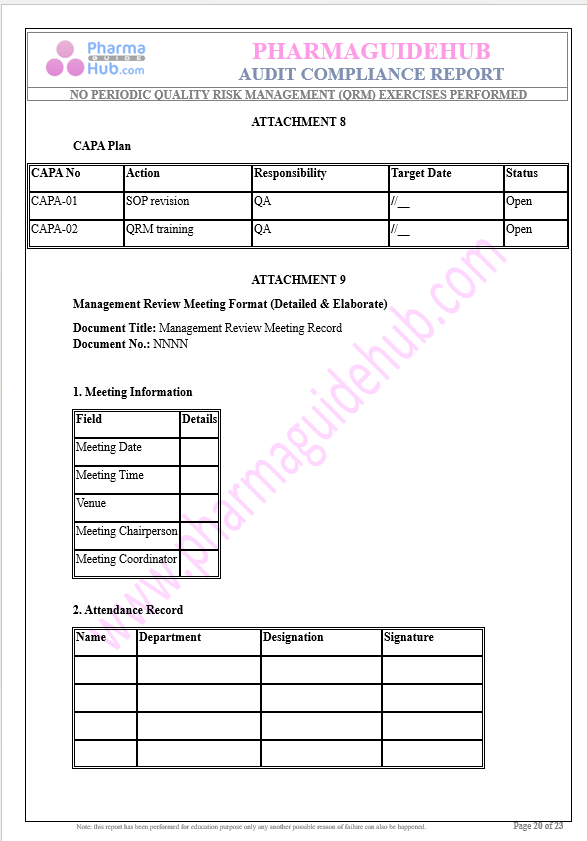
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
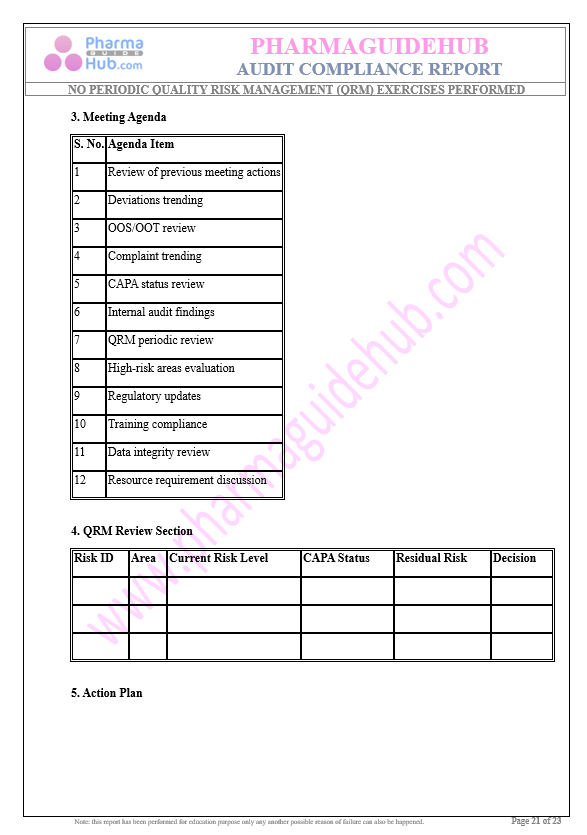
Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report

Key Parameter of the Below Page:
Audit Compliance Report
Find below pages for complete report
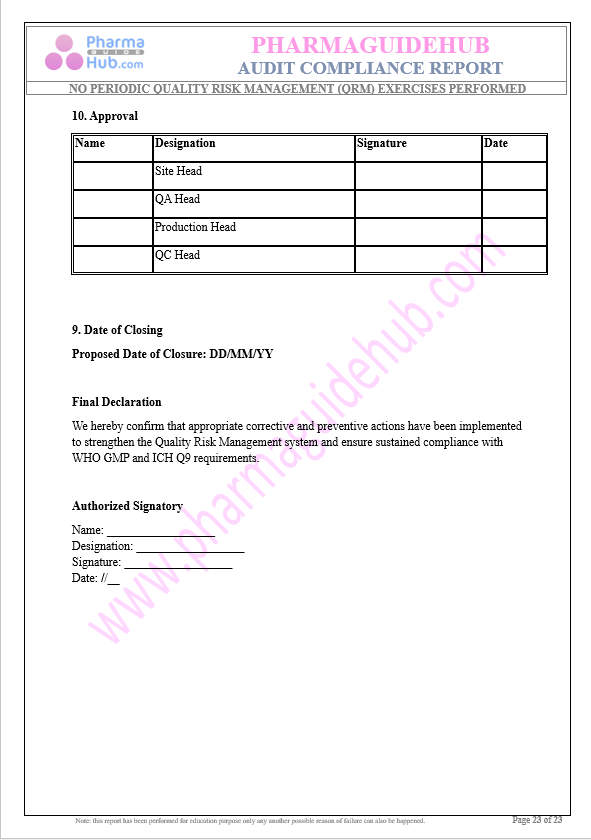
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/compliance-report-for-no-periodic-quality-risk-management-qrm-exercises-performed/




