OBJECTIVE:
To lay down the procedure for development, qualification, disqualification and requalification of suppliers for materials such as Pharmaceutical Aids, Solvents and Gases.
SCOPE:
This SOP is applicable to the development, qualification, disqualification and requalification of suppliers for materials such as Pharmaceutical Aids, Solvents and Gases which are routinely used during manufacturing operations at {Company Name} {Location}.
RESPONSIBILITY:
QA In-Charge/ Designee:
Requesting Purchase for identification of New Supplier and/or Alternate supplier for pharmaceutical aids, solvents and gases.
Initiating Non-Conformance as per SOP
Review of supplier provided investigation and CAPAs for observed Non-Conformances, initiate rejection of non-conforming material wherever required.
QC Executive/Designee:
Analysis / Evaluation of materials and preparation of reports.
ACCOUNTABILITY:
QA Head shall be Accountable for implementation of SOP.
PROCEDURE:
QA/ Formulation Research Development (FRD) shall provide material requirements and request Purchase to identify suppliers for New Materials. In case of Alternate/ Additional supplier development, Specifications shall be shared by Purchase with Suppliers.
Purchase shall identify suppliers and submit material requirements / specifications and request the supplier to share sample(s) [quantities as per evaluation requirement], standard specifications, testing procedures and Technical Data Sheet (TDS) / Certificate of analysis (COA) as applicable.
Supplier provided sample(s), TDS/COAs and standard specifications of material shall be submitted by Purchase to the Site QA.
Wherever sample(s) are required to be analysed, Quality Control and / or Formulation Analytical Research Development shall perform analysis, results of testing shall be compared with supplier TDS/COAs and the evaluation reports shall be shared with QA.
Raw materials such as gases, solvents etc. shall be accepted based on the supplier COAs/ TDS.QA personnel shall review all documentation (COA’s/TDS, trail reports, specific study reports etc.).
In charge Purchase/Designee shall forward Supplier Qualification Questionnaire to Supplier.
The Supplier Qualification Questionnaire consists of following;
Checklist: Checklist for Supplier Qualification.
Part A: Basic Information (Site details, contact numbers, Production capacity, turn over, Organization performance, major customers etc.).
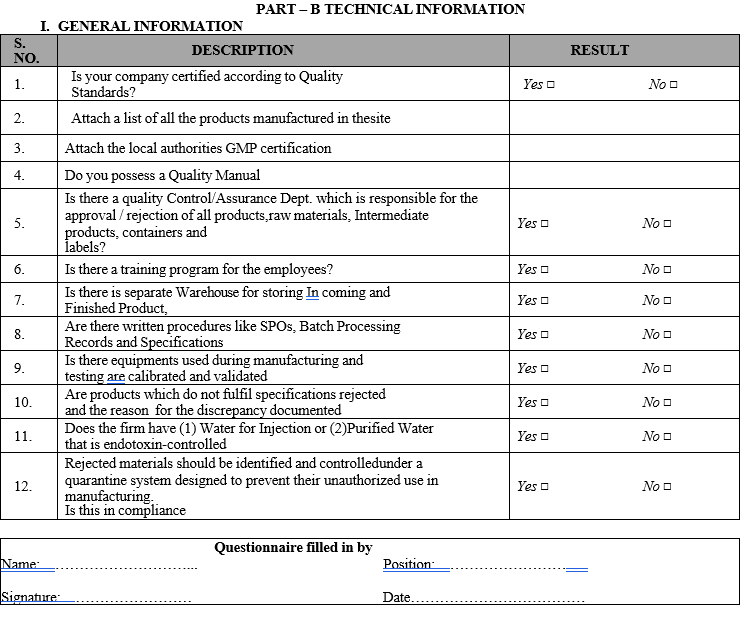
Part B: Technical Information of the Manufacturing site.
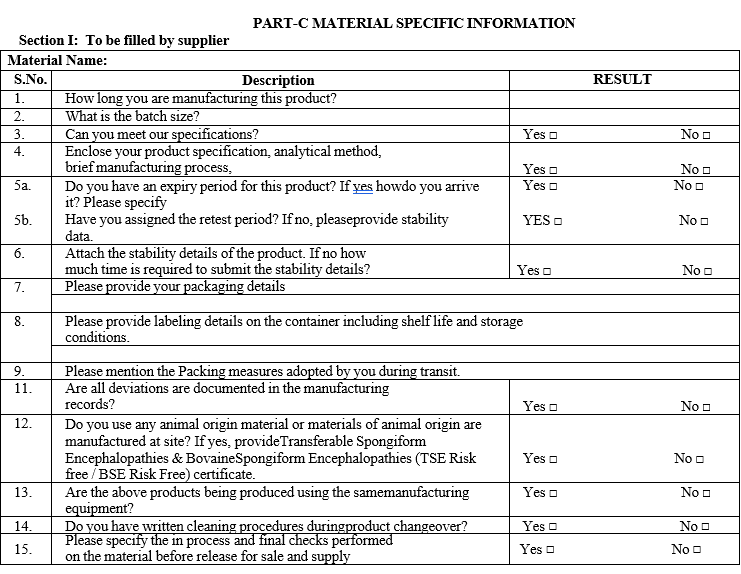
Part C: Material specific information
For a New Material, Purchase shall forward the Supplier Qualification Questionnaire to respective supplier as per the following criteria;

If standard information provided by supplier meets the requirement, then the same to be considered
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/
Checklist shall be forwarded to supplier for Solvents and Gases.
In case of pharmaceutical aids material wise Part C (Material specific information) will be prepared based on requirement, for documents Format VIII will be referred.
Suppliers shall fill relevant information and attach all supporting data and submit the same to purchase team personnel.
Filled questionnaire shall be submitted to QA.QA shall review available information (filled questionnaires, analytical data, trial reports, study reports etc.), if any additional information is required shall request supplier through purchase and receive the same.
QA shall assess all the information and determines a requirement for an Onsite Audit, as per SOP.
Wherever Onsite Supplier audits are required, same shall be completed.
Upon satisfactory review of information (Supplier Qualification Questionnaires Analytical data, Onsite audit reports, etc.) supplier shall be “Approved”.
Suppliers shall be “Conditionally Approved” when supplier qualification process is satisfactorily completed; however certain information is partly received and/or in scenarios of long lead time for procurement of the supplies.
Conditional approval of the supplier in respective warehouses shall be provided by QA by changing status of supplier to “New”.
Upon receipt of complete information, such conditionally approved supplier’s status shall be “Approved”.
The List of Approved Suppliers shall be available in QA department.
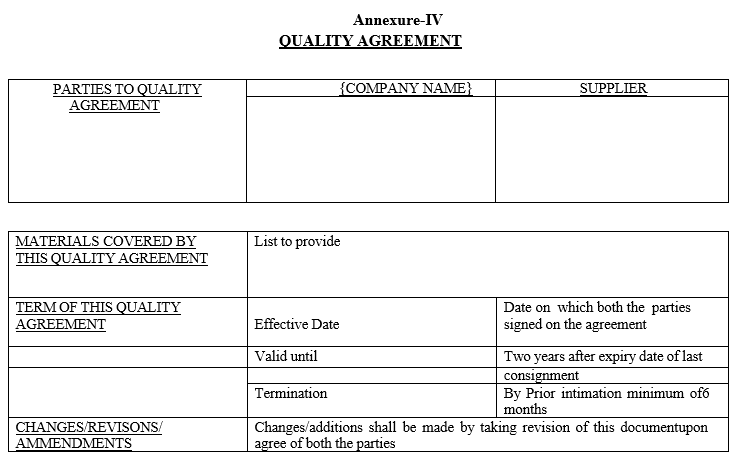
Wherever required “Quality Agreement” shall be prepared and approved before initiating routine supplies from suppliers.
Maintenance of Qualification Status:
Qualification status shall be maintained by;
Ensuring that all changes post approval of suppliers for commercial supplies follow a standard change management system as per SOP.
Periodic re-qualification of the suppliers for all high risk materials such as Sterile Pharmaceutical Aids, Solvents, Gases etc. once every 4 yrs. ±2 Months.
Note: Vendors shall be re-qualified by updating Questionnaires (Part-A, B&C) along with the product declarations as applicable. Existing documents may be considered in case there are no changes.
Routine monitoring of supplies for conformance to set specifications and/or Non-Conformances (if any);
Any Non-Conformance (NC) observed shall be intimated to Supplier by QA through Purchase for necessary investigation and corrective action to avoid recurrence.
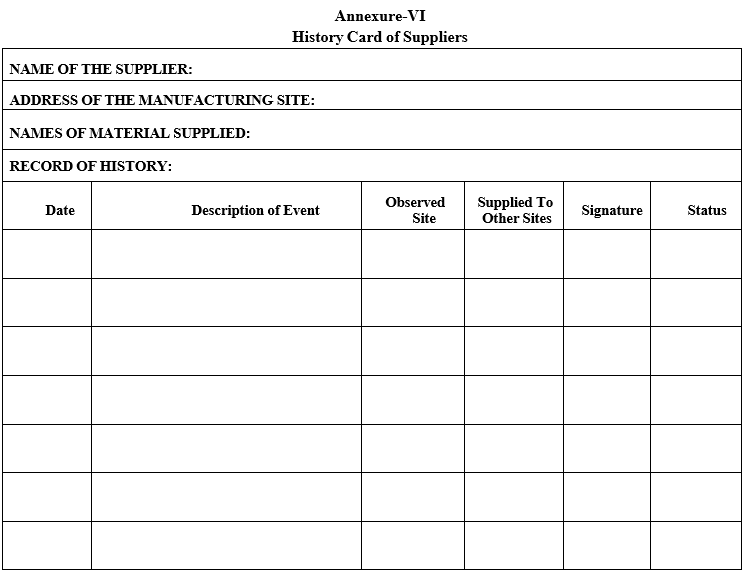
QA shall update the “History Card of Supplier” accordingly.
Based on Supplier investigation outcome if required onsite audit shall be planned.
During Non-Conformance evaluation, if impacting lot(s) material(s) consumption has to stopped, such material(s) shall be changed to “Hold”. Impacting lots shall be “Quarantined” until receipt of investigation report, highlighting corrective action plan from the supplier. Note: If alternative automated solutions for material handling and status indication, such as barcode systems material will not be kept Under Quarantine area.
If Procurement of material(s) from supplier(s) is to be stopped, QA shall change supplier status “Approved” to “Debarred” as per SOP.
Disqualification and Requalification of Suppliers:
A final approved supplier (Qualified for commercial supplies) shall be considered for disqualification based on any one or combination of the following;
Supply of contaminated material (such as Microbial contamination, mix up with different material etc.) which have adverse impact on product quality.
Three consecutive batches of material failed to meet defined specifications.
Abnormal Trend of NC’s including Quality Non-Impacting NC and Analytical Results observed during periodic requalification.
Inability to comply with gaps observed during site audits or any serious observation(s) having impact on product quality.
Non-compliance to Quality Agreements such as lack of communications of regulatory audit findings, regulatory actions if any, changes done without prior intimation etc.
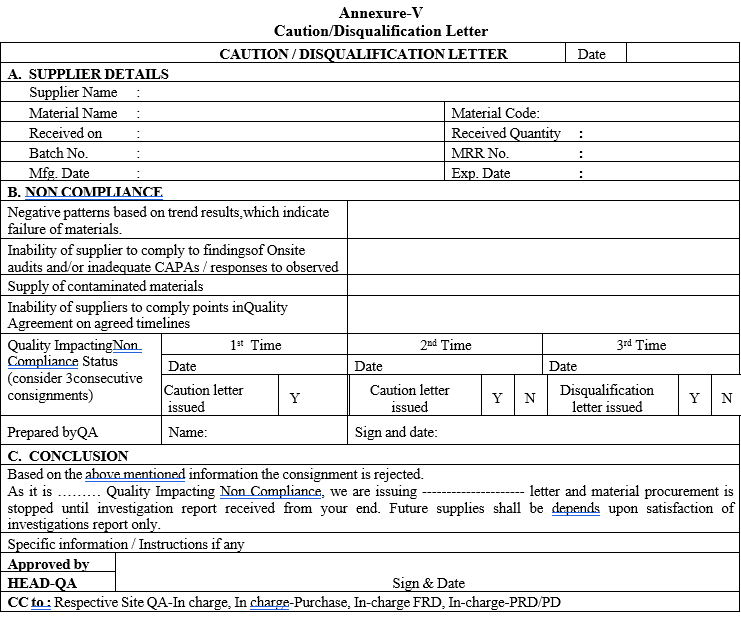
For all the above a prior intimation to supplier shall be given through a Caution letter and the response shall be evaluated before considering for disqualification of the supplier.
Supplier shall be disqualified, if the responses provided are inadequate by issuing a “Disqualification letter”.
A notification shall be issued by QA to respective Sites and other concerned and Supplier, through Purchase.
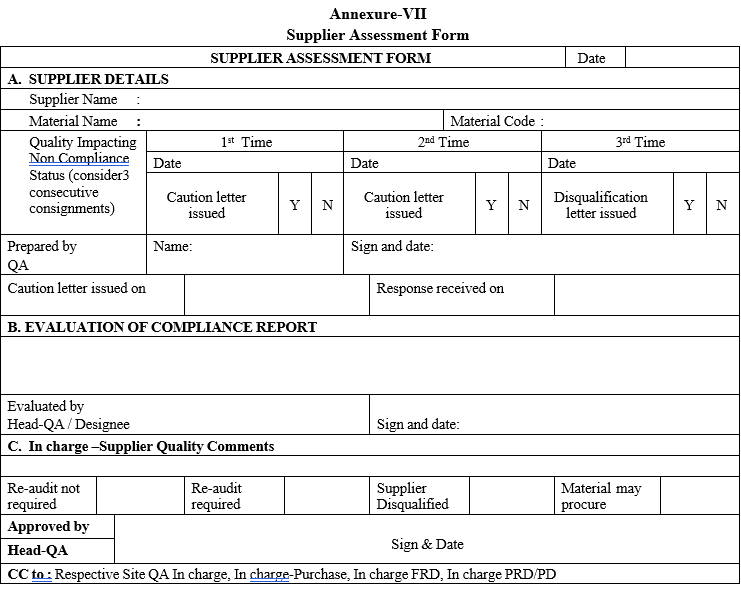
Disqualified supplier shall be considered for re-qualification only after thorough evaluation of supplier Quality systems, details shall be documented in “Supplier Assessment Form”.
The supplier status shall be changed from “Debarred” to “Approved” as per SOP.
The extent of disqualification of a supplier/material shall be determined based on following criteria;
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/
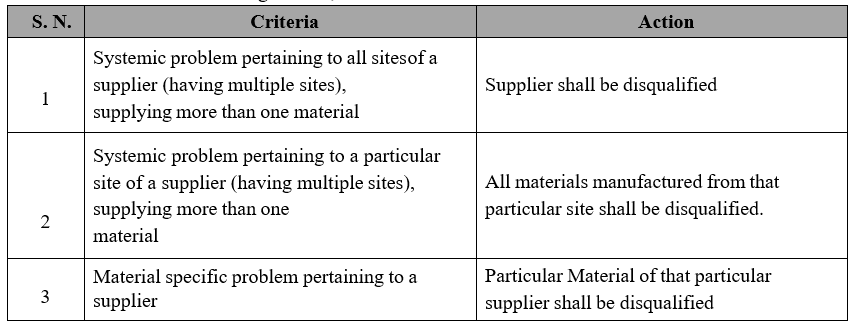
Qualified / approved suppliers from whom materials have not been sourced for longer periods (within periodic re-qualification frequency), re-initiation of supplies from such suppliers shall be considered.
REFERENCES:
Not Applicable
ANNEXURES:
| ANNEXURE NO. | TITLE OF ANNEXURE |
| Annexure-I | Checklist for Supplier Qualification of Solvents and Gases |
| Annexure-II | Supplier Qualification Questionnaire |
| Annexure-III | List of Approved Suppliers |
| Annexure-IV | Quality Agreement |
| Annexure-V | Caution/Disqualification Letter |
| Annexure-VI | History Card of Suppliers |
| Annexure-VII | Supplier Assessment Form |
| Annexure-VIII | List of Pharmaceuticals Aids and documents required for qualification process |
ENCLOSURES: SOP Training Record.
DISTRIBUTION:
Master Copy : Quality Assurance Department
Controlled Copy No. 01 : Head Quality Assurance
Controlled Copy No. 02 : Head Warehouse
Controlled Copy No. 03 : Head Quality control
Controlled Copy No. 04 : Head Engineering
ABBREVIATIONS:
| TDS | : | Technical Data Sheet |
| No. | : | Number |
| SOP | : | Standard Operating Procedure |
| MNC | : | Material Non Conformance |
| CAPA | : | Corrective And Preventive Actions |
| NC | : | Non Conformance |
| CC | : | Change Control |
| QA | : | Quality Assurance |
REVISION HISTORY:
CHANGE HISTORY LOG
| Revision No. | Details of Changes | Reason for Change | Effective Date |
| 00 | New SOP | Not Applicable |
Flow Chart
Annexure-I
Checklist for Supplier Qualification of Solvents and Gases
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/
Annexure-II
SUPPLIER QUALIFICATION QUESTIONNAIRE
PART-A GENERAL INFORMATION
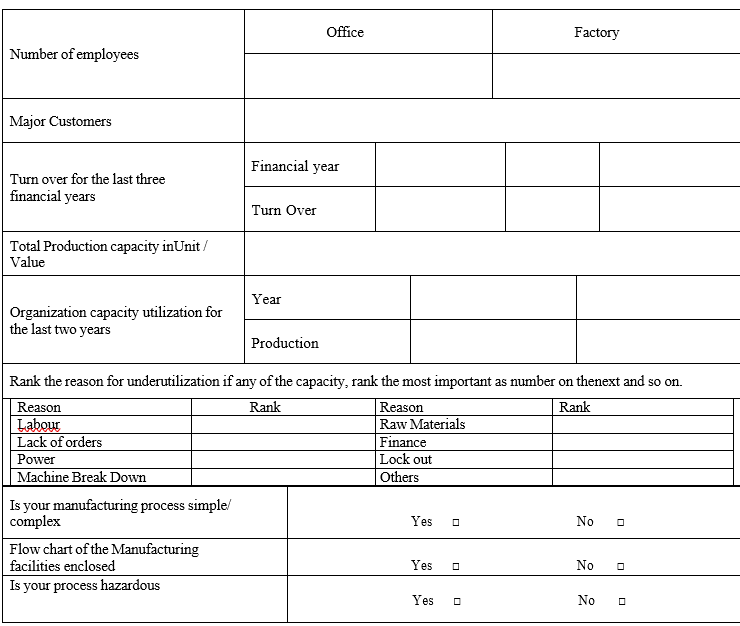
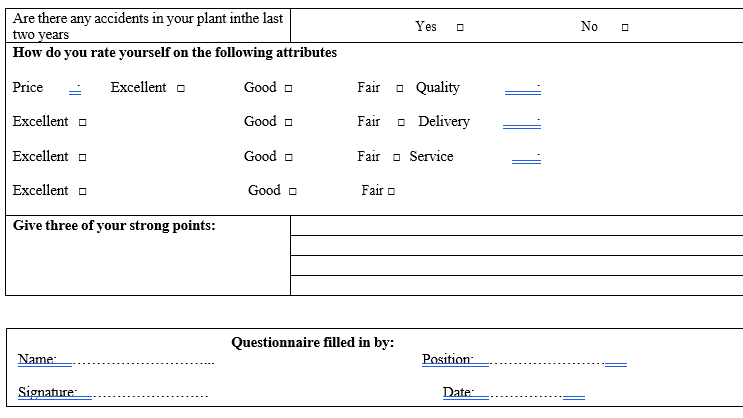
PART – B TECHNICAL INFORMATION

PART-C MATERIAL SPECIFIC INFORMATION
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/

PART C1 SUPPLIER QUALIFICATION QUESTIONNAIRE
DECLARATION OF ELEMENTAL ANALYSIS & METAL RESIDUES EVALUATION FOR EUROPE MARKET (APPLICABLE FOR SOLVENTS AND GASES)
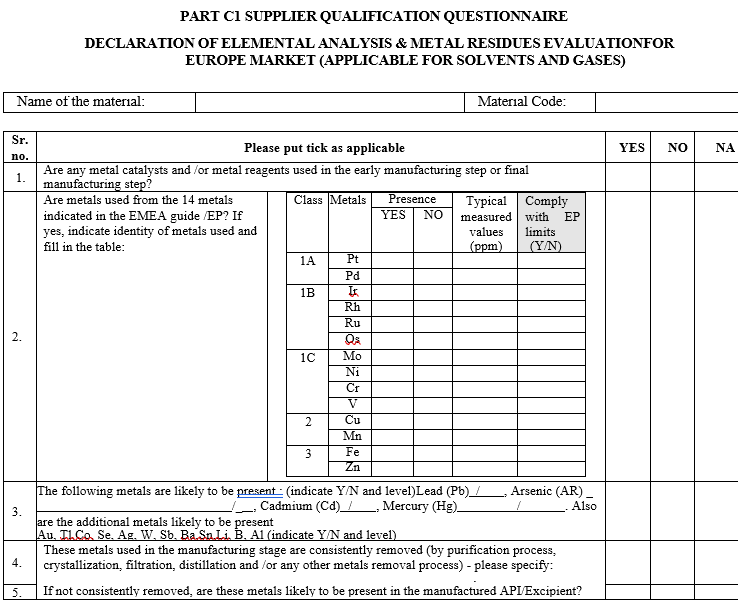

Part- C2 – SUPPLIER QUALIFICATION QUESTIONNAIRE
DECLARATION OF ELEMENTAL ANALYSIS & METAL RESIDUES EVALUATION FOR US MARKET (APPLICABLE FOR SOLVENTS AND GASES)
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/

Annexure-III
List of Approved Suppliers
QUALITY AGREEMENT
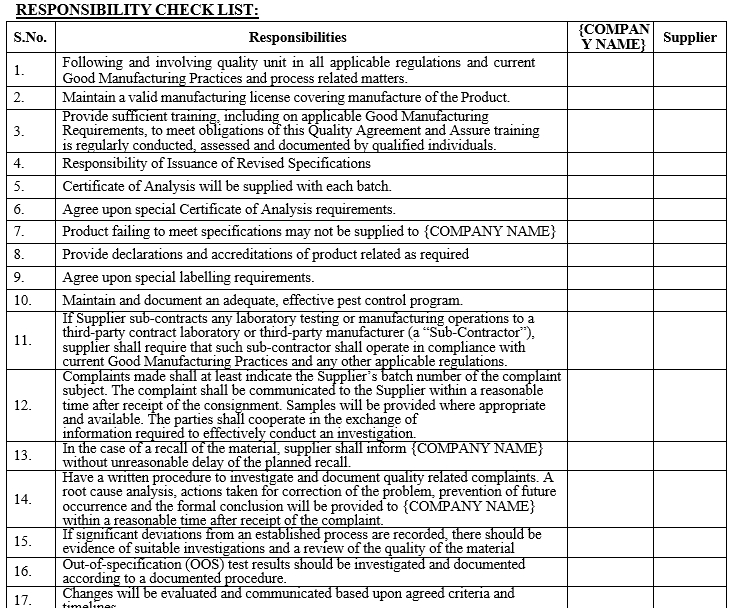
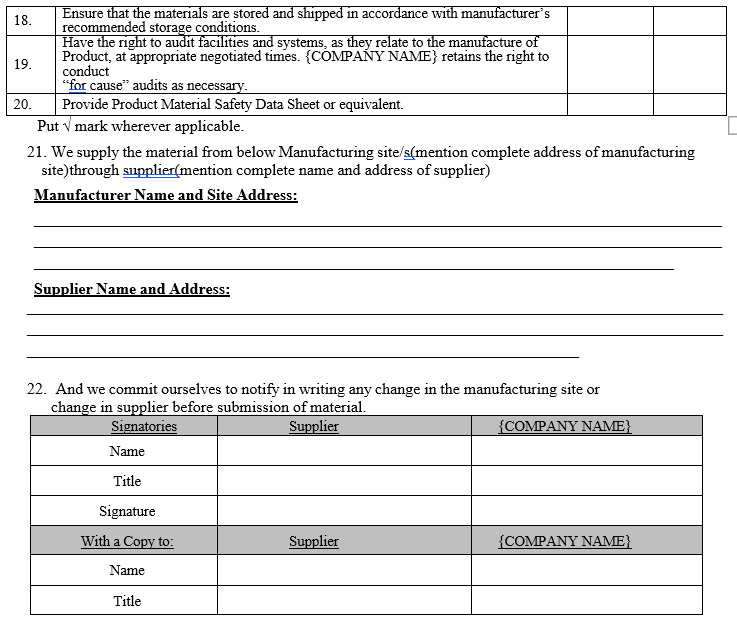
Annexure-IV
QUALITY AGREEMENT
Annexure-V
Caution/Disqualification Letter
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/
Annexure-VI
History Card of Suppliers
Annexure-VII
Supplier Assessment Form
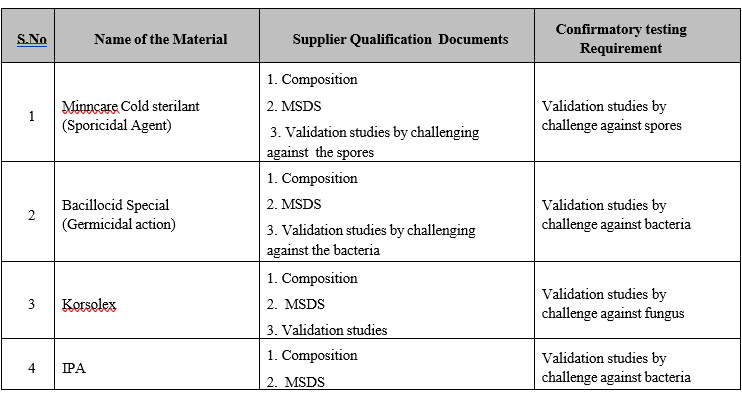
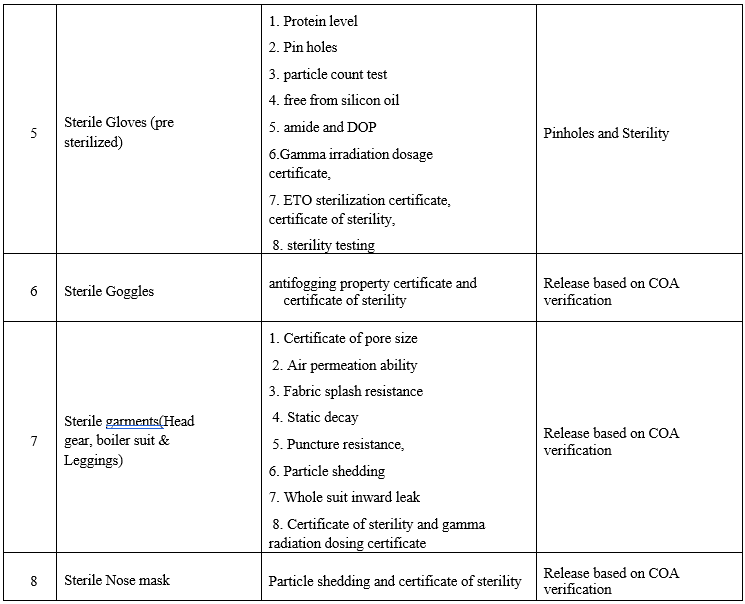
Annexure-VIII
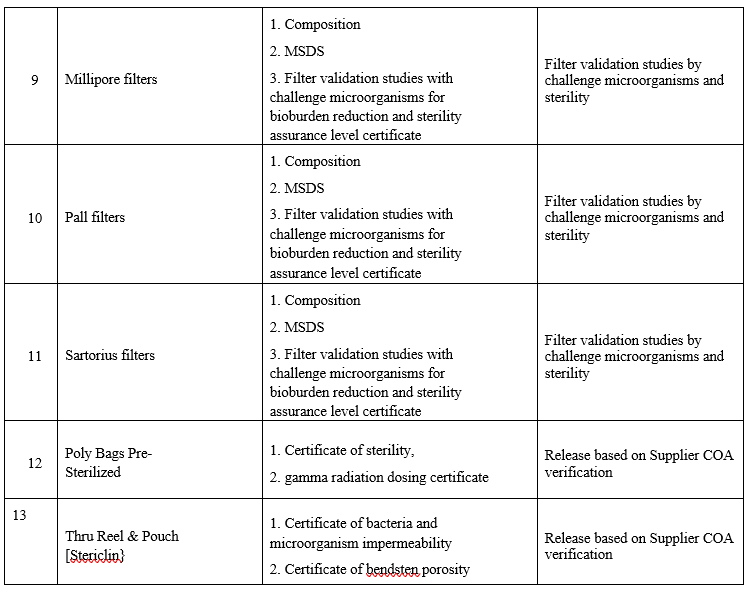
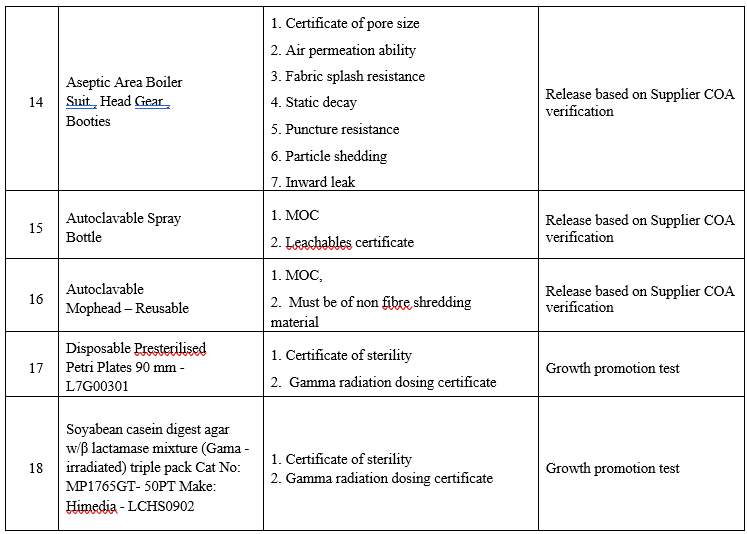
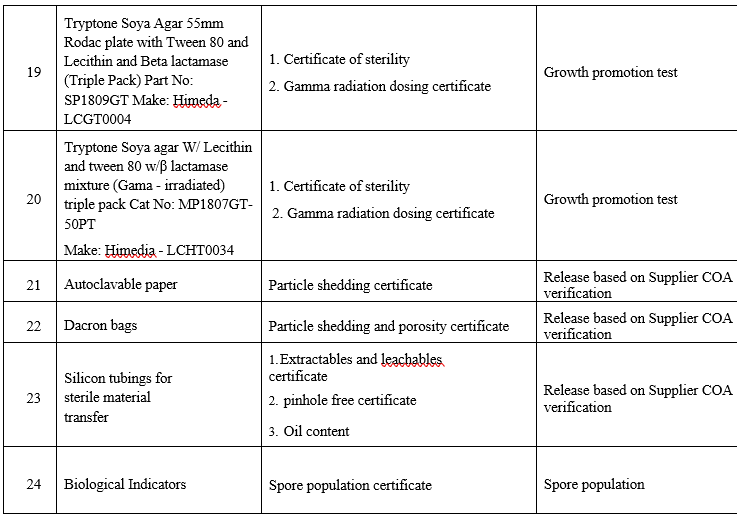
LIST OF PHARMACEUTICAL AIDS AND DOCUMENTS REQUIRED FOR QUALIFICATION PROCESS
Click the link to download word file copy of this document:
https://pharmaguidehub.com/product/supplier-qualification-program-for-pharmaceutical-aids-solvents-and-gases/





This SOP outlines a comprehensive process for supplier management, ensuring quality and compliance in the procurement of pharmaceutical aids, solvents, and gases. It emphasizes the importance of thorough evaluation and qualification of suppliers to maintain high standards in manufacturing operations. The procedure includes steps for identifying new or alternate suppliers, handling non-conformances, and preparing quality agreements. Regular re-qualification of suppliers is also mandated to ensure ongoing compliance. How does this SOP ensure consistency in supplier quality over time? Given the growing economic instability due to the events in the Middle East, many businesses are looking for guaranteed fast and secure payment solutions. Recently,
This document outlines a comprehensive procedure for managing suppliers of materials like Pharmaceutical Aids, Solvents, and Gases. It ensures that suppliers meet specific criteria and maintain quality standards. The process includes initial qualification, ongoing evaluation, and re-qualification of suppliers. Non-conformances are addressed through investigations and CAPAs to maintain material quality. Are there any specific criteria for immediate disqualification of a supplier in case of severe non-conformances?
This SOP outlines a comprehensive process for managing supplier qualifications, ensuring quality and compliance in pharmaceutical manufacturing. It emphasizes the importance of thorough evaluation and documentation for new and alternate suppliers. The role of the QA Head and Purchase team is crucial in maintaining standards and addressing non-conformances. Periodic re-qualification ensures continuous adherence to quality requirements. How does this SOP address potential risks associated with supplier changes in critical materials?